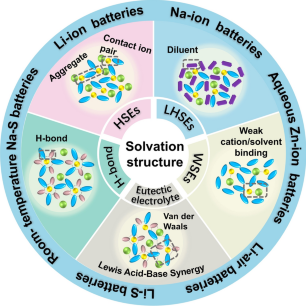
Electrolyte Evolution: A Roadmap from Solvation Structure to Next-Generation Batteries
Corresponding Author: Kunjie Zhu
Nano-Micro Letters,
Vol. 18 (2026), Article Number: 275
Abstract
Driven by global strategies for decarbonization and carbon neutrality, renewable-energy intermittency underscores the importance of large-scale electrochemical energy storage (EES). Rechargeable batteries, as the core components within EES, have long been restricted by limitations intrinsic to conventional dilute electrolytes, including narrow electrochemical stability windows, poor low-temperature performance, high flammability, and weak compatibility with high-voltage electrodes. Regulation of solvation structure in electrolytes has emerged as a key approach to overcome these bottlenecks. This review highlights five representative strategies: highly concentrated electrolytes, localized high-concentration electrolytes, weakly solvating electrolytes, hydrogen-bond regulated electrolytes, and eutectic electrolytes. These strategies have greatly advanced Li-ion, Na-ion, Zn-ion, Li–S, Li–air, and Na–S batteries. Finally, challenges ahead and opportunities in solvation-structure design are summarized to guide innovative and sustainable progress in next-generation energy storage technologies.
Highlights:
1 This review elucidates how innovative electrolytes (highly concentrated electrolytes, localized high-concentration electrolytes, etc.) reshape ion–solvent interactions.
2 Solvation-structure regulation is highlighted as the key to enhanced battery performance, with its recent advances summarized across diverse battery systems.
3 This review outlines challenges and opportunities in solvation-structure design to guide next-generation energy storage technologies.
Keywords
Download Citation
Endnote/Zotero/Mendeley (RIS)BibTeX
- R. Santhanam, B. Rambabu, Research progress in high voltage spinel LiNi0.5Mn1.5O4 material. J. Power. Sources 195(17), 5442–5451 (2010). https://doi.org/10.1016/j.jpowsour.2010.03.067
- K. Kanamura, Anodic oxidation of nonaqueous electrolytes on cathode materials and current collectors for rechargeable lithium batteries. J. Power. Sources 81, 123–129 (1999). https://doi.org/10.1016/S0378-7753(99)00141-X
- D. Di Lecce, J. Manzi, F.M. Vitucci, A. De Bonis, S. Panero et al., Effect of the iron doping in LiCoPO4 cathode materials for lithium cells. Electrochim. Acta 185, 17–27 (2015). https://doi.org/10.1016/j.electacta.2015.10.107
- L. Chen, X. Fan, E. Hu, X. Ji, J. Chen et al., Achieving high energy density through increasing the output voltage: a highly reversible 5.3 V battery. Chem 5(4), 896–912 (2019). https://doi.org/10.1016/j.chempr.2019.02.003
- L. Bai, Y. Xu, Y. Liu, D. Zhang, S. Zhang et al., Metal-organic framework glass stabilizes high-voltage cathodes for efficient lithium-metal batteries. Nat. Commun. 16, 3484 (2025). https://doi.org/10.1038/s41467-025-58639-z
- B. Lee, E. Paek, D. Mitlin, S.W. Lee, Sodium metal anodes: emerging solutions to dendrite growth. Chem. Rev. 119(8), 5416–5460 (2019). https://doi.org/10.1021/acs.chemrev.8b00642
- A. von Wald Cresce, O. Borodin, K. Xu, Correlating Li+ solvation sheath structure with interphasial chemistry on graphite. J. Phys. Chem. C 116(50), 26111–26117 (2012). https://doi.org/10.1021/jp303610t
- X. Guo, H. Xie, Z. Ma, P. Kumar, Z. Zhang et al., Electrolyte exchange experiment in batteries: failure analysis and prospect. J. Energy Chem. 103, 601–623 (2024). https://doi.org/10.1016/j.jechem.2024.12.026
- L. Suo, O. Borodin, T. Gao, M. Olguin, J. Ho et al., “Water-in-salt” electrolyte enables high-voltage aqueous lithium-ion chemistries. Science 350(6263), 938–943 (2015). https://doi.org/10.1126/science.aab1595
- M. Moshkovich, M. Cojocaru, H.E. Gottlieb, D. Aurbach, The study of the anodic stability of alkyl carbonate solutions by in situ FTIR spectroscopy, EQCM, NMR and MS. J. Electroanal. Chem. 497(1–2), 84–96 (2001). https://doi.org/10.1016/s0022-0728(00)00457-5
- A. Gupta, A. Manthiram, Designing advanced lithium-based batteries for low-temperature conditions. Adv. Energy Mater. 10(38), 2001972 (2020). https://doi.org/10.1002/aenm.202001972
- J. Wang, Y. Yamada, K. Sodeyama, E. Watanabe, K. Takada et al., Fire-extinguishing organic electrolytes for safe batteries. Nat. Energy 3(1), 22–29 (2018). https://doi.org/10.1038/s41560-017-0033-8
- C. Li, C. Ni, X.-L. Huang, L. Yao, L. Yang et al., Recent advances and perspectives of electrolyte additives for enhanced anode reversibility in aqueous zinc-ion batteries. Chem. Commun. 61(58), 10643–10667 (2025). https://doi.org/10.1039/d5cc02302e
- J.-W. Zhang, J.-L. Sun, D.-N. Zhao, Y.-J. Zhao, X.-Y. Hu et al., Tuning solvation structure to enhance low temperature kinetics of lithium-ion batteries. Energy Storage Mater. 72, 103698 (2024). https://doi.org/10.1016/j.ensm.2024.103698
- Z. Yu, H. Wang, X. Kong, W. Huang, Y. Tsao et al., Molecular design for electrolyte solvents enabling energy-dense and long-cycling lithium metal batteries. Nat. Energy 5(7), 526–533 (2020). https://doi.org/10.1038/s41560-020-0634-5
- S. Chen, J. Zheng, D. Mei, K.S. Han, M.H. Engelhard et al., High-voltage lithium-metal batteries enabled by localized high-concentration electrolytes. Adv. Mater. 30(21), 1706102 (2018). https://doi.org/10.1002/adma.201706102
- D.M. Seo, O. Borodin, S.-D. Han, P.D. Boyle, W.A. Henderson, Electrolyte solvation and ionic association II. Acetonitrile-lithium salt mixtures: highly dissociated salts. J. Electrochem. Soc. 159(9), A1489–A1500 (2012). https://doi.org/10.1149/2.035209jes
- Y. Yamada, A. Yamada, Superconcentrated electrolytes for lithium batteries. J. Electrochem. Soc. 162(14), A2406–A2423 (2015). https://doi.org/10.1149/2.0041514jes
- Y. Yamada, K. Furukawa, K. Sodeyama, K. Kikuchi, M. Yaegashi et al., Unusual stability of acetonitrile-based superconcentrated electrolytes for fast-charging lithium-ion batteries. J. Am. Chem. Soc. 136(13), 5039–5046 (2014). https://doi.org/10.1021/ja412807w
- T. Li, X.-Q. Zhang, P. Shi, Q. Zhang, Fluorinated solid-electrolyte interphase in high-voltage lithium metal batteries. Joule 3(11), 2647–2661 (2019). https://doi.org/10.1016/j.joule.2019.09.022
- L. Suo, O. Borodin, W. Sun, X. Fan, C. Yang et al., Advanced high-voltage aqueous lithium-ion battery enabled by “water-in-bisalt” electrolyte. Angew. Chem. Int. Ed. 55(25), 7136–7141 (2016). https://doi.org/10.1002/anie.201602397
- T. Jin, X. Ji, P.F. Wang, K. Zhu, J. Zhang et al., High‐energy aqueous sodium‐ion batteries. Angew. Chem. Int. Ed. 60(21), 11943–11948 (2021). https://doi.org/10.1002/anie.202017167
- L. Jiang, Y. Lu, C. Zhao, L. Liu, J. Zhang et al., Building aqueous K-ion batteries for energy storage. Nat. Energy 4, 495–503 (2019). https://doi.org/10.1038/s41560-019-0388-0
- J. Wang, Y. Yamada, K. Sodeyama, C.H. Chiang, Y. Tateyama et al., Superconcentrated electrolytes for a high-voltage lithium-ion battery. Nat. Commun. 7, 12032 (2016). https://doi.org/10.1038/ncomms12032
- L. Suo, W. Xue, M. Gobet, S.G. Greenbaum, C. Wang et al., Fluorine-donating electrolytes enable highly reversible 5-V-class Li metal batteries. Proc. Natl. Acad. Sci. U. S. A. 115(6), 1156–1161 (2018). https://doi.org/10.1073/pnas.1712895115
- L. Xing, X. Zheng, M. Schroeder, J. Alvarado, A. von Wald Cresce et al., Deciphering the ethylene carbonate–propylene carbonate mystery in Li-ion batteries. Acc. Chem. Res. 51(2), 282–289 (2018). https://doi.org/10.1021/acs.accounts.7b00474
- S.-K. Jeong, M. Inaba, Y. Iriyama, T. Abe, Z. Ogumi, Electrochemical intercalation of lithium ion within graphite from propylene carbonate solutions. Electrochem. Solid-State Lett. 6(1), A13 (2003). https://doi.org/10.1149/1.1526781
- T. Doi, R. Masuhara, M. Hashinokuchi, Y. Shimizu, M. Inaba, Concentrated LiPF6/PC electrolyte solutions for 5-V LiNi0.5Mn1.5O4 positive electrode in lithium-ion batteries. Electrochim. Acta 209, 219–224 (2016). https://doi.org/10.1016/j.electacta.2016.05.062
- X. Li, X. Ou, Y. Tang, 6.0 V high-voltage and concentrated electrolyte toward high energy density K-based dual-graphite battery. Adv. Energy Mater. 10(41), 2002567 (2020). https://doi.org/10.1002/aenm.202002567
- K. Sodeyama, Y. Yamada, K. Aikawa, A. Yamada, Y. Tateyama, Sacrificial anion reduction mechanism for electrochemical stability improvement in highly concentrated Li-salt electrolyte. J. Phys. Chem. C 118(26), 14091–14097 (2014). https://doi.org/10.1021/jp501178n
- X. Fan, L. Chen, X. Ji, T. Deng, S. Hou et al., Highly fluorinated interphases enable high-voltage Li-metal batteries. Chem 4(1), 174–185 (2018). https://doi.org/10.1016/j.chempr.2017.10.017
- S. Liu, J. Mao, L. Zhang, W.K. Pang, A. Du et al., Manipulating the solvation structure of nonflammable electrolyte and interface to enable unprecedented stability of graphite anodes beyond 2 years for safe potassium-ion batteries. Adv. Mater. 33, 2006313 (2021). https://doi.org/10.1002/adma.202006313
- Y. Yamada, K. Usui, C.H. Chiang, K. Kikuchi, K. Furukawa et al., General observation of lithium intercalation into graphite in ethylene-carbonate-free superconcentrated electrolytes. ACS Appl. Mater. Interfaces 6(14), 10892–10899 (2014). https://doi.org/10.1021/am5001163
- Y. Yamada, Developing new functionalities of superconcentrated electrolytes for lithium-ion batteries. Electrochemistry 85(9), 559–565 (2017). https://doi.org/10.5796/electrochemistry.85.559
- W. Liu, J. Li, W. Li, H. Xu, C. Zhang et al., Inhibition of transition metals dissolution in cobalt-free cathode with ultrathin robust interphase in concentrated electrolyte. Nat. Commun. 11, 3629 (2020). https://doi.org/10.1038/s41467-020-17396-x
- S. Jiao, X. Ren, R. Cao, M.H. Engelhard, Y. Liu et al., Stable cycling of high-voltage lithium metal batteries in ether electrolytes. Nat. Energy 3(9), 739–746 (2018). https://doi.org/10.1038/s41560-018-0199-8
- X. Ren, L. Zou, S. Jiao, D. Mei, M.H. Engelhard et al., High-concentration ether electrolytes for stable high-voltage lithium metal batteries. ACS Energy Lett. 4(4), 896–902 (2019). https://doi.org/10.1021/acsenergylett.9b00381
- S.-K. Jeong, H.-Y. Seo, D.-H. Kim, H.-K. Han, J.-G. Kim et al., Suppression of dendritic lithium formation by using concentrated electrolyte solutions. Electrochem. Commun. 10(4), 635–638 (2008). https://doi.org/10.1016/j.elecom.2008.02.006
- S. Wang, Y. Ding, G. Zhou, G. Yu, A. Manthiram, Durability of the Li1+xTi2–xAlx(PO4)3 solid electrolyte in lithium–sulfur batteries. ACS Energy Lett. 1(6), 1080–1085 (2016). https://doi.org/10.1021/acsenergylett.6b00481
- L. Wang, Y. Qu, C. Su, R. Li, D. Liu et al., Interfacial engineering of Li anode and Ni-rich cathode via anion-regulated SEI/CEI layers in flame-retardant nanofiber-reinforced polymer electrolyte for lithium metal batteries. Adv. Energy Mater. 15(35), 2501185 (2025). https://doi.org/10.1002/aenm.202501185
- Z. Wang, H. Tu, X. Ma, S. Lu, G. Su et al., Flame-retardant electrolytes with electrochemically-inert and weakly coordinating dichloroalkane diluents for practical lithium metal batteries. Nat. Commun. 16, 10188 (2025). https://doi.org/10.1038/s41467-025-65138-8
- K. Lee, S.-H. Kwon, J. Kim, E. Park, I. Kim et al., Fluorinated cyclic ether diluent for high-voltage lithium metal batteries. ACS Energy Lett. 9(5), 2201–2211 (2024). https://doi.org/10.1021/acsenergylett.4c00481
- X. Wang, Y. Bai, Z. Lv, Y. Liu, W. Dong et al., In situ polymerized localized high-concentration electrolytes for ultrahigh-rate sodium metal batteries. Adv. Energy Mater. 15(28), 2500887 (2025). https://doi.org/10.1002/aenm.202500887
- D. Guo, J. Wang, Z. Cui, Z. Shi, G. Henkelman et al., Low-temperature sodium–sulfur batteries enabled by ionic liquid in localized high concentration electrolytes. Adv. Funct. Mater. 34(51), 2409494 (2024). https://doi.org/10.1002/adfm.202409494
- Q. Wu, Y. Qi, Revealing heterogeneous electric double layer (EDL) structures of localized high-concentration electrolytes (LHCEs) and their impact on solid–electrolyte interphase (SEI) formation in lithium batteries. Energy Environ. Sci. 18(6), 3036–3046 (2025). https://doi.org/10.1039/d5ee00206k
- Y. Chen, Z. Yu, P. Rudnicki, H. Gong, Z. Huang et al., Steric effect tuned ion solvation enabling stable cycling of high-voltage lithium metal battery. J. Am. Chem. Soc. 143(44), 18703–18713 (2021). https://doi.org/10.1021/jacs.1c09006
- J. Choi, H. Jeong, J. Jang, A.-R. Jeon, I. Kang et al., Weakly solvating solution enables chemical prelithiation of graphite–SiOx anodes for high-energy Li-ion batteries. J. Am. Chem. Soc. 143(24), 9169–9176 (2021). https://doi.org/10.1021/jacs.1c03648
- J. Holoubek, H. Liu, Z. Wu, Y. Yin, X. Xing et al., Tailoring electrolyte solvation for Li metal batteries cycled at ultra-low temperature. Nat. Energy 6(3), 303–313 (2021). https://doi.org/10.1038/s41560-021-00783-z
- Y.-X. Yao, X. Chen, C. Yan, X.-Q. Zhang, W.-L. Cai et al., Regulating interfacial chemistry in lithium-ion batteries by a weakly solvating electrolyte. Angew. Chem. Int. Ed. 60(8), 4090–4097 (2021). https://doi.org/10.1002/anie.202011482
- M. Mao, X. Ji, Q. Wang, Z. Lin, M. Li et al., Anion-enrichment interface enables high-voltage anode-free lithium metal batteries. Nat. Commun. 14, 1082 (2023). https://doi.org/10.1038/s41467-023-36853-x
- X. Zheng, Z. Gu, J. Fu, H. Wang, X. Ye et al., Knocking down the kinetic barriers towards fast-charging and low-temperature sodium metal batteries. Energy Environ. Sci. 14(9), 4936–4947 (2021). https://doi.org/10.1039/d1ee01404h
- S. Zhu, J. Chen, Dual strategy with Li-ion solvation and solid electrolyte interphase for high Coulombic efficiency of lithium metal anode. Energy Storage Mater. 44, 48–56 (2022). https://doi.org/10.1016/j.ensm.2021.10.007
- H.M. Law, Z. Wang, S. Xu, L. Shen, B. Py et al., A weakly solvating solvent-based quasi-solid electrolyte for sodium metal batteries. Energy Environ. Sci. 18(19), 8838–8848 (2025). https://doi.org/10.1039/d5ee02153g
- T. Jin, X.-Y. Li, M. Zhao, S. Feng, Z. Li et al., Promoting the rate performances of weakly solvating electrolyte-based lithium‒sulfur batteries. Angew. Chem. Int. Ed. 64(32), e202504898 (2025). https://doi.org/10.1002/anie.202504898
- Z.-X. Chen, J.-J. Zhao, G.-Y. Fang, S. Zhang, J. Ma et al., Accelerating the sulfur redox kinetics in weakly-solvating lithium‒sulfur batteries under lean-electrolyte conditions. Energy Storage Mater. 83, 104502 (2025). https://doi.org/10.1016/j.ensm.2025.104502
- D. Feng, Y. Xie, Y. Jiao, P. Wu, Leveraging cation effect for low temperature aqueous Zn-based batteries. Nat. Commun. 16, 9254 (2025). https://doi.org/10.1038/s41467-025-64278-1
- N. Chang, T. Li, R. Li, S. Wang, Y. Yin et al., An aqueous hybrid electrolyte for low-temperature zinc-based energy storage devices. Energy Environ. Sci. 13(10), 3527–3535 (2020). https://doi.org/10.1039/d0ee01538e
- J. Liu, C. Yang, X. Chi, B. Wen, W. Wang et al., Water/sulfolane hybrid electrolyte achieves ultralow-temperature operation for high-voltage aqueous lithium-ion batteries. Adv. Funct. Mater. 32, 2106811 (2022). https://doi.org/10.1002/adfm.202106811
- P. Wang, T.C. Li, Y. Liu, C. Lin, Y. Cui et al., Targeted docking of localized hydrogen bond for efficient and reversible zinc-ion batteries. Angew. Chem. Int. Ed. 64(15), e202422547 (2025). https://doi.org/10.1002/anie.202422547
- J. Hao, L. Yuan, C. Ye, D. Chao, K. Davey et al., Boosting zinc electrode reversibility in aqueous electrolytes by using low-cost antisolvents. Angew. Chem. Int. Ed. 60(13), 7366–7375 (2021). https://doi.org/10.1002/anie.202016531
- Q. Zhang, Y. Ma, Y. Lu, L. Li, F. Wan et al., Modulating electrolyte structure for ultralow temperature aqueous zinc batteries. Nat. Commun. 11, 4463 (2020). https://doi.org/10.1038/s41467-020-18284-0
- L. Cao, D. Li, F.A. Soto, V. Ponce, B. Zhang et al., Highly reversible aqueous zinc batteries enabled by zincophilic–zincophobic interfacial layers and interrupted hydrogen-bond electrolytes. Angew. Chem. Int. Ed. 60(34), 18845–18851 (2021). https://doi.org/10.1002/anie.202107378
- A. von Wald Cresce, M. Gobet, O. Borodin, J. Peng, S.M. Russell et al., Anion solvation in carbonate-based electrolytes. J. Phys. Chem. C 119(49), 27255–27264 (2015). https://doi.org/10.1021/acs.jpcc.5b08895
- K.W. Schroder, A.G. Dylla, L.D.C. Bishop, E.R. Kamilar, J. Saunders et al., Effects of solute–solvent hydrogen bonding on nonaqueous electrolyte structure. J. Phys. Chem. Lett. 6(15), 2888–2891 (2015). https://doi.org/10.1021/acs.jpclett.5b01216
- X. Zhang, X. Dong, X. Yue, J. Gao, Z. Shi et al., Solvation regulation via hydrogen bonding to mitigate Al current collector corrosion for high-voltage Li-ion batteries. Adv. Energy Mater. 15(10), 2403588 (2025). https://doi.org/10.1002/aenm.202403588
- Z. Yang, J. He, W.-H. Lai, J. Peng, X.-H. Liu et al., Fire-retardant, stable-cycling and high-safety sodium ion battery. Angew. Chem. Int. Ed. 60(52), 27086–27094 (2021). https://doi.org/10.1002/anie.202112382
- Z. Yang, J. He, W.H. Lai, J. Peng, X.H. Liu et al., Fire-retardant, Stable-cycling and high-safety sodium ion battery. Angew. Chem. Int. Ed. 133(52), 27292–27300 (2021). https://doi.org/10.1002/anie.202112382
- J.A.P. Coutinho, S.P. Pinho, Special issue on deep eutectic solvents: a foreword. Fluid Phase Equilib. 448, 1 (2017). https://doi.org/10.1016/j.fluid.2017.06.011
- L. Geng, X. Wang, K. Han, P. Hu, L. Zhou et al., Eutectic electrolytes in advanced metal-ion batteries. ACS Energy Lett. 7(1), 247–260 (2022). https://doi.org/10.1021/acsenergylett.1c02088
- C. Zhang, L. Zhang, G. Yu, Eutectic electrolytes as a promising platform for next-generation electrochemical energy storage. Acc. Chem. Res. 53(8), 1648–1659 (2020). https://doi.org/10.1021/acs.accounts.0c00360
- X. Lu, E.J. Hansen, G. He, J. Liu, Eutectic electrolytes chemistry for rechargeable Zn batteries. Small 18(21), 2200550 (2022). https://doi.org/10.1002/smll.202200550
- J. Wu, Q. Liang, X. Yu, Q.-F. Lü, L. Ma et al., Deep eutectic solvents for boosting electrochemical energy storage and conversion: a review and perspective. Adv. Funct. Mater. 31(22), 2011102 (2021). https://doi.org/10.1002/adfm.202011102
- B.B. Hansen, S. Spittle, B. Chen, D. Poe, Y. Zhang et al., Deep eutectic solvents: a review of fundamentals and applications. Chem. Rev. 121(3), 1232–1285 (2021). https://doi.org/10.1021/acs.chemrev.0c00385
- M.E. Di Pietro, A. Mele, Deep eutectics and analogues as electrolytes in batteries. J. Mol. Liq. 338, 116597 (2021). https://doi.org/10.1016/j.molliq.2021.116597
- E.L. Smith, A.P. Abbott, K.S. Ryder, Deep eutectic solvents (DESs) and their applications. Chem. Rev. 114(21), 11060–11082 (2014). https://doi.org/10.1021/cr300162p
- M. Fang, X. Yue, Y. Dong, Y. Chen, Z. Liang, A temperature-dependent solvating electrolyte for wide-temperature and fast-charging lithium metal batteries. Joule 8(1), 91–103 (2024). https://doi.org/10.1016/j.joule.2023.12.012
- J. Zhao, J. Zhang, W. Yang, B. Chen, Z. Zhao et al., Water-in-deep eutectic solvent” electrolytes enable zinc metal anodes for rechargeable aqueous batteries. Nano Energy 57, 625–634 (2019). https://doi.org/10.1016/j.nanoen.2018.12.086
- W. Yang, X. Du, J. Zhao, Z. Chen, J. Li et al., Hydrated eutectic electrolytes with ligand-oriented solvation shells for long-cycling zinc-organic batteries. Joule 4(7), 1557–1574 (2020). https://doi.org/10.1016/j.joule.2020.05.018
- Y. Hu, Novel room temperature molten salt electrolyte based on LiTFSI and acetamide for lithium batteries. Electrochem. Commun. 6(1), 28–32 (2004). https://doi.org/10.1016/j.elecom.2003.10.009
- A. Boisset, J. Jacquemin, M. Anouti, Physical properties of a new deep eutectic solvent based on lithium bis [(trifluoromethyl)sulfonyl] imide and N-methylacetamide as superionic suitable electrolyte for lithium ion batteries and electric double layer capacitors. Electrochim. Acta 102, 120–126 (2013). https://doi.org/10.1016/j.electacta.2013.03.150
- A. Boisset, S. Menne, J. Jacquemin, A. Balducci, M. Anouti, Deep eutectic solvents based on N-methylacetamide and a lithium salt as suitable electrolytes for lithium-ion batteries. Phys. Chem. Chem. Phys. 15(46), 20054–20063 (2013). https://doi.org/10.1039/c3cp53406e
- Z. Hu, F. Xian, Z. Guo, C. Lu, X. Du et al., Nonflammable nitrile deep eutectic electrolyte enables high-voltage lithium metal batteries. Chem. Mater. 32(8), 3405–3413 (2020). https://doi.org/10.1021/acs.chemmater.9b05003
- Y. Lu, Z. Tu, L.A. Archer, Stable lithium electrodeposition in liquid and nanoporous solid electrolytes. Nat. Mater. 13(10), 961–969 (2014). https://doi.org/10.1038/nmat4041
- J. Qian, W.A. Henderson, W. Xu, P. Bhattacharya, M. Engelhard et al., High rate and stable cycling of lithium metal anode. Nat. Commun. 6, 6362 (2015). https://doi.org/10.1038/ncomms7362
- C. Guo, K. Zhang, Q. Zhao, L. Pei, J. Chen, High-performance sodium batteries with the 9, 10-anthraquinone/CMK-3 cathode and an ether-based electrolyte. Chem. Commun. 51(50), 10244–10247 (2015). https://doi.org/10.1039/c5cc02251g
- R. Cao, K. Mishra, X. Li, J. Qian, M.H. Engelhard et al., Enabling room temperature sodium metal batteries. Nano Energy 30, 825–830 (2016). https://doi.org/10.1016/j.nanoen.2016.09.013
- L. Schafzahl, I. Hanzu, M. Wilkening, S.A. Freunberger, An electrolyte for reversible cycling of sodium metal and intercalation compounds. Chemsuschem 10(2), 401–408 (2017). https://doi.org/10.1002/cssc.201601222
- I.A. Shkrob, T.W. Marin, Y. Zhu, D.P. Abraham, Why bis(fluorosulfonyl)imide is a “magic anion” for electrochemistry. J. Phys. Chem. C 118(34), 19661–19671 (2014). https://doi.org/10.1021/jp506567p
- J. Lee, Y. Lee, J. Lee, S.-M. Lee, J.-H. Choi et al., Ultraconcentrated sodium bis(fluorosulfonyl)imide-based electrolytes for high-performance sodium metal batteries. ACS Appl. Mater. Interfaces 9(4), 3723–3732 (2017). https://doi.org/10.1021/acsami.6b14878
- M. He, K.C. Lau, X. Ren, N. Xiao, W.D. McCulloch et al., Concentrated electrolyte for the sodium–oxygen battery: solvation structure and improved cycle life. Angew. Chem. Int. Ed. 55(49), 15310–15314 (2016). https://doi.org/10.1002/anie.201608607
- X. Shi, J. Xie, J. Wang, S. Xie, Z. Yang et al., A weakly solvating electrolyte towards practical rechargeable aqueous zinc-ion batteries. Nat. Commun. 15, 302 (2024). https://doi.org/10.1038/s41467-023-44615-y
- T. Li, H. Yang, X. Dong, H. Ma, J. Cai et al., Co-regulation of interface and bulk for enhanced localized high-concentration electrolytes in stable and practical zinc metal batteries. Angew. Chem. Int. Ed. 64(29), e202501183 (2025). https://doi.org/10.1002/anie.202501183
- C. Huang, M. Zhao, C. Xu, Y. Lv, M. Fang et al., Engineering anion-diluent matrix for ion-decoupled localized high-concentration electrolytes toward highly stable aqueous zinc ion batteries. Angew. Chem. Int. Ed. 64(40), e202511410 (2025). https://doi.org/10.1002/anie.202511410
- Y. Yamada, Y. Takazawa, K. Miyazaki, T. Abe, Electrochemical lithium intercalation into graphite in dimethyl sulfoxide-based electrolytes: effect of solvation structure of lithium ion. J. Phys. Chem. C 114(26), 11680–11685 (2010). https://doi.org/10.1021/jp1037427
- F. Li, L. Kong, Y. Sun, Y. Jin, P. Hou, Micron-sized monocrystalline LiNi1/3Co1/3Mn1/3O2 as high-volumetric-energy-density cathode for lithium-ion batteries. J. Mater. Chem. A 6, 12344–12352 (2018). https://doi.org/10.1039/c8ta03363c
- A. Manthiram, A reflection on lithium-ion battery cathode chemistry. Nat. Commun. 11, 1550 (2020). https://doi.org/10.1038/s41467-020-15355-0
- B. Wang, H.A. Doan, S.-B. Son, D.P. Abraham, S.E. Trask et al., Data-driven design of electrolyte additives supporting high-performance 5 V LiNi0.5Mn1.5O4 positive electrodes. Nat. Commun. 16, 3413 (2025). https://doi.org/10.1038/s41467-025-57961-w
- Z. Zhang, M. Avdeev, H. Chen, W. Yin, W.H. Kan et al., Lithiated Prussian blue analogues as positive electrode active materials for stable non-aqueous lithium-ion batteries. Nat. Commun. 13, 7790 (2022). https://doi.org/10.1038/s41467-022-35376-1
- C.S. Johnson, J.-S. Kim, C. Lefief, N. Li, J.T. Vaughey et al., The significance of the Li2MnO3 component in ‘composite’ xLi2MnO3·(1–x)LiMn0.5Ni0.5O2 electrodes. Electrochem. Commun. 6(10), 1085–1091 (2004). https://doi.org/10.1016/j.elecom.2004.08.002
- D. Aurbach, B.D. McCloskey, L.F. Nazar, P.G. Bruce, Advances in understanding mechanisms underpinning lithium–air batteries. Nat. Energy 1(9), 16128 (2016). https://doi.org/10.1038/nenergy.2016.128
- P. Hartmann, C.L. Bender, M. Vračar, A.K. Dürr, A. Garsuch et al., A rechargeable room-temperature sodium superoxide (NaO2) battery. Nat. Mater. 12(3), 228–232 (2013). https://doi.org/10.1038/nmat3486
- Z. Peng, S.A. Freunberger, L.J. Hardwick, Y. Chen, V. Giordani et al., Oxygen reactions in a non-aqueous Li+ electrolyte. Angew. Chem. Int. Ed. 50(28), 6351–6355 (2011). https://doi.org/10.1002/anie.201100879
- Y.-C. Lu, B.M. Gallant, D.G. Kwabi, J.R. Harding, R.R. Mitchell et al., Lithium–oxygen batteries: bridging mechanistic understanding and battery performance. Energy Environ. Sci. 6(3), 750 (2013). https://doi.org/10.1039/c3ee23966g
- Z.-L. Wang, D. Xu, J.-J. Xu, X.-B. Zhang, Oxygen electrocatalysts in metal–air batteries: from aqueous to nonaqueous electrolytes. Chem. Soc. Rev. 43(22), 7746–7786 (2014). https://doi.org/10.1039/c3cs60248f
- S.A. Freunberger, Y. Chen, Z. Peng, J.M. Griffin, L.J. Hardwick et al., Reactions in the rechargeable lithium–O2 battery with alkyl carbonate electrolytes. J. Am. Chem. Soc. 133(20), 8040–8047 (2011). https://doi.org/10.1021/ja2021747
- C. Laoire, S. Mukerjee, E.J. Plichta, M.A. Hendrickson, K. Abraham, Rechargeable lithium/TEGDME-LiPF6/O2 battery. J. Electrochem. Soc. 158(3), A302–A308 (2011). https://doi.org/10.1149/1.3531981
- R.S. Assary, J. Lu, P. Du, X. Luo, X. Zhang et al., The effect of oxygen crossover on the anode of a Li–O2 battery using an ether-based solvent: insights from experimental and computational studies. ChemSusChem 6(1), 51–55 (2013). https://doi.org/10.1002/cssc.201200810
- C.O. Laoire, S. Mukerjee, K.M. Abraham, E.J. Plichta, M.A. Hendrickson, Influence of nonaqueous solvents on the electrochemistry of oxygen in the rechargeable lithium–air battery. J. Phys. Chem. C 114(19), 9178–9186 (2010). https://doi.org/10.1021/jp102019y
- D. Xu, Z.-L. Wang, J.-J. Xu, L.-L. Zhang, L.-M. Wang et al., A stable sulfone based electrolyte for high performance rechargeable Li–O2 batteries. Chem. Commun. 48(95), 11674 (2012). https://doi.org/10.1039/c2cc36815c
- B. Sun, C. Pompe, S. Dongmo, J. Zhang, K. Kretschmer et al., Challenges for developing rechargeable room-temperature sodium oxygen batteries. Adv. Mater. Technol. 3(9), 1800110 (2018). https://doi.org/10.1002/admt.201800110
- Y. Wang, Y. Wang, Y.-X. Wang, X. Feng, W. Chen et al., Developments and perspectives on emerging high-energy-density sodium-metal batteries. Chem 5(10), 2547–2570 (2019). https://doi.org/10.1016/j.chempr.2019.05.026
- J. Lu, Y. Jung Lee, X. Luo, K. Chun Lau, M. Asadi et al., A lithium–oxygen battery based on lithium superoxide. Nature 529(7586), 377–382 (2016). https://doi.org/10.1038/nature16484
- W.A. Henderson, F. McKenna, M.A. Khan, N.R. Brooks, V.G. Young Jr. et al., Glyme–Lithium Bis(trifluoromethanesulfonyl)imide and Glyme–Lithium Bis(perfluoroethanesulfonyl)imide phase behavior and solvate structures. Chem. Mater. 17(9), 2284–2289 (2005). https://doi.org/10.1021/cm047881j
- Y. Liu, L. Suo, H. Lin, W. Yang, Y. Fang et al., Novel approach for a high-energy-density Li–air battery: tri-dimensional growth of Li2O2 crystals tailored by electrolyte Li+ ion concentrations. J. Mater. Chem. A 2(24), 9020–9024 (2014). https://doi.org/10.1039/c4ta00834k
- B. Liu, W. Xu, P. Yan, X. Sun, M.E. Bowden et al., Enhanced cycling stability of rechargeable Li–O2 batteries using high-concentration electrolytes. Adv. Funct. Mater. 26(4), 605–613 (2016). https://doi.org/10.1002/adfm.201503697
- N. Tachikawa, K. Yamauchi, E. Takashima, J.-W. Park, K. Dokko et al., Reversibility of electrochemical reactions of sulfur supported on inverse opal carbon in glyme–Li salt molten complex electrolytes. Chem. Commun. 47(28), 8157–8159 (2011). https://doi.org/10.1039/c1cc12415c
- X.-Y. Li, M. Zhao, Y.-W. Song, C.-X. Bi, Z. Li et al., Polysulfide chemistry in metal–sulfur batteries. Chem. Soc. Rev. 54(10), 4822–4873 (2025). https://doi.org/10.1039/d4cs00318g
- L. Suo, Y.-S. Hu, H. Li, M. Armand, L. Chen, A new class of solvent-in-salt electrolyte for high-energy rechargeable metallic lithium batteries. Nat. Commun. 4, 1481 (2013). https://doi.org/10.1038/ncomms2513
- K. Dokko, N. Tachikawa, K. Yamauchi, M. Tsuchiya, A. Yamazaki et al., Solvate ionic liquid electrolyte for Li–S batteries. J. Electrochem. Soc. 160(8), A1304–A1310 (2013). https://doi.org/10.1149/2.111308jes
- M. Cuisinier, P.-E. Cabelguen, B.D. Adams, A. Garsuch, M. Balasubramanian et al., Unique behaviour of nonsolvents for polysulphides in lithium–sulphur batteries. Energy Environ. Sci. 7(8), 2697–2705 (2014). https://doi.org/10.1039/c4ee00372a
- J. Zheng, X. Fan, G. Ji, H. Wang, S. Hou et al., Manipulating electrolyte and solid electrolyte interphase to enable safe and efficient Li–S batteries. Nano Energy 50, 431–440 (2018). https://doi.org/10.1016/j.nanoen.2018.05.065
- X.-Y. Li, B.-Q. Li, S. Feng, Z. Li, L. Shen et al., Two-stage solvation of lithium polysulfides in working lithium–sulfur batteries. J. Am. Chem. Soc. 147(18), 15435–15447 (2025). https://doi.org/10.1021/jacs.5c01588
- Q. Pang, X. Liang, C.Y. Kwok, L.F. Nazar, Advances in lithium–sulfur batteries based on multifunctional cathodes and electrolytes. Nat. Energy 1(9), 16132 (2016). https://doi.org/10.1038/nenergy.2016.132
- Y. Yamada, M. Yaegashi, T. Abe, A. Yamada, A superconcentrated ether electrolyte for fast-charging Li-ion batteries. Chem. Commun. 49(95), 11194 (2013). https://doi.org/10.1039/c3cc46665e
- K. Yoshida, M. Nakamura, Y. Kazue, N. Tachikawa, S. Tsuzuki et al., Oxidative-stability enhancement and charge transport mechanism in glyme–lithium salt equimolar complexes. J. Am. Chem. Soc. 133(33), 13121–13129 (2011). https://doi.org/10.1021/ja203983r
- F. Li, T. Zhang, Y. Yamada, A. Yamada, H. Zhou, Enhanced cycling performance of Li-O2 batteries by the optimized electrolyte concentration of LiTFSA in glymes. Adv. Energy Mater. 3(4), 532–538 (2013). https://doi.org/10.1002/aenm.201200776
- K. Matsumoto, K. Inoue, K. Nakahara, R. Yuge, T. Noguchi et al., Suppression of aluminum corrosion by using high concentration LiTFSI electrolyte. J. Power. Sources 231, 234–238 (2013). https://doi.org/10.1016/j.jpowsour.2012.12.028
- L. Suo, Y.-S. Hu, H. Li, M. Armand, L. Chen, A new class of solvent-in-salt electrolyte for high-energy rechargeable metallic lithium batteries. Nat. Commun. 4, 1481 (2013). https://doi.org/10.1038/ncomms2513
- S.-I. Okuoka, Y. Ogasawara, Y. Suga, M. Hibino, T. Kudo et al., A new sealed lithium-peroxide battery with a Co-doped Li2O cathode in a superconcentrated lithium bis(fluorosulfonyl)amide electrolyte. Sci. Rep. 4, 5684 (2014). https://doi.org/10.1038/srep05684
- C. Zhang, A. Yamazaki, J. Murai, J.-W. Park, T. Mandai et al., Chelate effects in glyme/lithium bis(trifluoromethanesulfonyl)amide solvate ionic liquids, part 2: importance of solvate-structure stability for electrolytes of lithium batteries. J. Phys. Chem. C 118(31), 17362–17373 (2014). https://doi.org/10.1021/jp504099q
- H. Kim, F. Wu, J.T. Lee, N. Nitta, H.-T. Lin et al., In situ formation of protective coatings on sulfur cathodes in lithium batteries with LiFSI-based organic electrolytes. Adv. Energy Mater. 5(6), 1401792 (2015). https://doi.org/10.1002/aenm.201401792
- R. Petibon, C.P. Aiken, L. Ma, D. Xiong, J.R. Dahn, The use of ethyl acetate as a sole solvent in highly concentrated electrolyte for Li-ion batteries. Electrochim. Acta 154, 287–293 (2015). https://doi.org/10.1016/j.electacta.2014.12.093
- Y. Yamada, C.H. Chiang, K. Sodeyama, J. Wang, Y. Tateyama et al., Corrosion prevention mechanism of aluminum metal in superconcentrated electrolytes. ChemElectroChem 2(11), 1687–1694 (2015). https://doi.org/10.1002/celc.201500235
- J. Alvarado, M.A. Schroeder, M. Zhang, O. Borodin, E. Gobrogge et al., A carbonate-free, sulfone-based electrolyte for high-voltage Li-ion batteries. Mater. Today 21(4), 341–353 (2018). https://doi.org/10.1016/j.mattod.2018.02.005
- Z. Zeng, V. Murugesan, K.S. Han, X. Jiang, Y. Cao et al., Non-flammable electrolytes with high salt-to-solvent ratios for Li-ion and Li-metal batteries. Nat. Energy 3(8), 674–681 (2018). https://doi.org/10.1038/s41560-018-0196-y
- Z. Wang, Y. Sun, Y. Mao, F. Zhang, L. Zheng et al., Highly concentrated dual-anion electrolyte for non-flammable high-voltage Li-metal batteries. Energy Storage Mater. 30, 228–237 (2020). https://doi.org/10.1016/j.ensm.2020.05.020
- M. Zhao, G. Xu, D. Lu, B. Xie, L. Huang et al., Formulating a non-flammable highly concentrated dual-salt electrolyte for wide temperature high-nickel lithium ion batteries. J. Electrochem. Soc. 168(5), 050511 (2021). https://doi.org/10.1149/1945-7111/abfb39
- W. Cai, Y. Deng, Z. Deng, Y. Jia, Z. Li et al., Quasi-localized high-concentration electrolytes for high-voltage lithium metal batteries. Adv. Energy Mater. 13(31), 2301396 (2023). https://doi.org/10.1002/aenm.202301396
- F. Hai, Y. Yi, Z. Xiao, J. Guo, X. Gao et al., A low-cost, fluorine-free localized highly concentrated electrolyte toward ultra-high loading lithium metal batteries. Adv. Energy Mater. 14(17), 2304253 (2024). https://doi.org/10.1002/aenm.202304253
- Z. Han, L. Chen, G. Zheng, D. Zhang, K. Yang et al., Ionic liquid reinforcing ether coordination of localized high concentration electrolyte enables high-voltage lithium metal batteries. Adv. Mater. 37(14), 2416668 (2025). https://doi.org/10.1002/adma.202416668
- J. Peng, H. Zhang, Z. Zeng, H. Zhang, H. Pei et al., Tailoring solvation structures via precise diluent engineering for high-rate 500 wh kg–1 lithium-metal batteries. Adv. Mater. 37(45), e09109 (2025). https://doi.org/10.1002/adma.202509109
- T. Zhang, J. Yu, T. Lin, Y. Song, M. Li et al., Fluorinated deep eutectic gel electrolytes for sustainable lithium metal batteries. J. Am. Chem. Soc. 147(36), 32861–32872 (2025). https://doi.org/10.1021/jacs.5c08642
- C. Luo, C. Ning, X. Huang, P. Zhang, L. Yang et al., Molecular engineering of weakly solvating dinitrile electrolytes for long-lasting room-temperature lithium metal batteries. Angew. Chem. Int. Ed. 64(29), e202507051 (2025). https://doi.org/10.1002/anie.202507051
- H. Yang, H. Li, J. Li, Z. Sun, K. He et al., The rechargeable aluminum battery: opportunities and challenges. Angew. Chem. Int. Ed. 58(35), 11978–11996 (2019). https://doi.org/10.1002/anie.201814031
- E. Miele, W.M. Dose, I. Manyakin, M.H. Frosz, Z. Ruff et al., Hollow-core optical fibre sensors for operando Raman spectroscopy investigation of Li-ion battery liquid electrolytes. Nat. Commun. 13, 1651 (2022). https://doi.org/10.1038/s41467-022-29330-4
- Y.-H. Wang, S. Zheng, W.-M. Yang, R.-Y. Zhou, Q.-F. He et al., In situ Raman spectroscopy reveals the structure and dissociation of interfacial water. Nature 600, 81–85 (2021). https://doi.org/10.1038/s41586-021-04068-z
- Y. Yamada, J. Wang, S. Ko, E. Watanabe, A. Yamada, Advances and issues in developing salt-concentrated battery electrolytes. Nat. Energy 4(4), 269–280 (2019). https://doi.org/10.1038/s41560-019-0336-z
- N. Yao, X. Chen, Z.-H. Fu, Q. Zhang, Applying classical, Ab initio, and machine-learning molecular dynamics simulations to the liquid electrolyte for rechargeable batteries. Chem. Rev. 122(12), 10970–11021 (2022). https://doi.org/10.1021/acs.chemrev.1c00904
- H. Wang, L. Zhang, J. Han, E. Weinan, DeePMD-kit: a deep learning package for many-body potential energy representation and molecular dynamics. Comput. Phys. Commun. 228, 178–184 (2018). https://doi.org/10.1016/j.cpc.2018.03.016
- D. Lu, H. Wang, M. Chen, L. Lin, R. Car et al., 86 PFLOPS deep potential molecular dynamics simulation of 100 million atoms with ab initio accuracy. Comput. Phys. Commun. 259, 107624 (2021). https://doi.org/10.1016/j.cpc.2020.107624
- Y. Hu, H. Su, J. Fu, J. Luo, Q. Yu et al., Solid solvation structure design improves all-solid-state organic batteries. Nat. Chem. 17(9), 1313–1322 (2025). https://doi.org/10.1038/s41557-025-01866-0
- Q. You, Y. Sun, F. Wang, J. Cheng, F. Tang, Decoding the competing effects of dynamic solvation structures on nuclear magnetic resonance chemical shifts of battery electrolytes via machine learning. J. Am. Chem. Soc. 147(17), 14667–14676 (2025). https://doi.org/10.1021/jacs.5c02710
- S. Chen, G. Wu, H. Jiang, J. Wang, T. Chen et al., External Li supply reshapes Li deficiency and lifetime limit of batteries. Nature 638(8051), 676–683 (2025). https://doi.org/10.1038/s41586-024-08465-y
References
R. Santhanam, B. Rambabu, Research progress in high voltage spinel LiNi0.5Mn1.5O4 material. J. Power. Sources 195(17), 5442–5451 (2010). https://doi.org/10.1016/j.jpowsour.2010.03.067
K. Kanamura, Anodic oxidation of nonaqueous electrolytes on cathode materials and current collectors for rechargeable lithium batteries. J. Power. Sources 81, 123–129 (1999). https://doi.org/10.1016/S0378-7753(99)00141-X
D. Di Lecce, J. Manzi, F.M. Vitucci, A. De Bonis, S. Panero et al., Effect of the iron doping in LiCoPO4 cathode materials for lithium cells. Electrochim. Acta 185, 17–27 (2015). https://doi.org/10.1016/j.electacta.2015.10.107
L. Chen, X. Fan, E. Hu, X. Ji, J. Chen et al., Achieving high energy density through increasing the output voltage: a highly reversible 5.3 V battery. Chem 5(4), 896–912 (2019). https://doi.org/10.1016/j.chempr.2019.02.003
L. Bai, Y. Xu, Y. Liu, D. Zhang, S. Zhang et al., Metal-organic framework glass stabilizes high-voltage cathodes for efficient lithium-metal batteries. Nat. Commun. 16, 3484 (2025). https://doi.org/10.1038/s41467-025-58639-z
B. Lee, E. Paek, D. Mitlin, S.W. Lee, Sodium metal anodes: emerging solutions to dendrite growth. Chem. Rev. 119(8), 5416–5460 (2019). https://doi.org/10.1021/acs.chemrev.8b00642
A. von Wald Cresce, O. Borodin, K. Xu, Correlating Li+ solvation sheath structure with interphasial chemistry on graphite. J. Phys. Chem. C 116(50), 26111–26117 (2012). https://doi.org/10.1021/jp303610t
X. Guo, H. Xie, Z. Ma, P. Kumar, Z. Zhang et al., Electrolyte exchange experiment in batteries: failure analysis and prospect. J. Energy Chem. 103, 601–623 (2024). https://doi.org/10.1016/j.jechem.2024.12.026
L. Suo, O. Borodin, T. Gao, M. Olguin, J. Ho et al., “Water-in-salt” electrolyte enables high-voltage aqueous lithium-ion chemistries. Science 350(6263), 938–943 (2015). https://doi.org/10.1126/science.aab1595
M. Moshkovich, M. Cojocaru, H.E. Gottlieb, D. Aurbach, The study of the anodic stability of alkyl carbonate solutions by in situ FTIR spectroscopy, EQCM, NMR and MS. J. Electroanal. Chem. 497(1–2), 84–96 (2001). https://doi.org/10.1016/s0022-0728(00)00457-5
A. Gupta, A. Manthiram, Designing advanced lithium-based batteries for low-temperature conditions. Adv. Energy Mater. 10(38), 2001972 (2020). https://doi.org/10.1002/aenm.202001972
J. Wang, Y. Yamada, K. Sodeyama, E. Watanabe, K. Takada et al., Fire-extinguishing organic electrolytes for safe batteries. Nat. Energy 3(1), 22–29 (2018). https://doi.org/10.1038/s41560-017-0033-8
C. Li, C. Ni, X.-L. Huang, L. Yao, L. Yang et al., Recent advances and perspectives of electrolyte additives for enhanced anode reversibility in aqueous zinc-ion batteries. Chem. Commun. 61(58), 10643–10667 (2025). https://doi.org/10.1039/d5cc02302e
J.-W. Zhang, J.-L. Sun, D.-N. Zhao, Y.-J. Zhao, X.-Y. Hu et al., Tuning solvation structure to enhance low temperature kinetics of lithium-ion batteries. Energy Storage Mater. 72, 103698 (2024). https://doi.org/10.1016/j.ensm.2024.103698
Z. Yu, H. Wang, X. Kong, W. Huang, Y. Tsao et al., Molecular design for electrolyte solvents enabling energy-dense and long-cycling lithium metal batteries. Nat. Energy 5(7), 526–533 (2020). https://doi.org/10.1038/s41560-020-0634-5
S. Chen, J. Zheng, D. Mei, K.S. Han, M.H. Engelhard et al., High-voltage lithium-metal batteries enabled by localized high-concentration electrolytes. Adv. Mater. 30(21), 1706102 (2018). https://doi.org/10.1002/adma.201706102
D.M. Seo, O. Borodin, S.-D. Han, P.D. Boyle, W.A. Henderson, Electrolyte solvation and ionic association II. Acetonitrile-lithium salt mixtures: highly dissociated salts. J. Electrochem. Soc. 159(9), A1489–A1500 (2012). https://doi.org/10.1149/2.035209jes
Y. Yamada, A. Yamada, Superconcentrated electrolytes for lithium batteries. J. Electrochem. Soc. 162(14), A2406–A2423 (2015). https://doi.org/10.1149/2.0041514jes
Y. Yamada, K. Furukawa, K. Sodeyama, K. Kikuchi, M. Yaegashi et al., Unusual stability of acetonitrile-based superconcentrated electrolytes for fast-charging lithium-ion batteries. J. Am. Chem. Soc. 136(13), 5039–5046 (2014). https://doi.org/10.1021/ja412807w
T. Li, X.-Q. Zhang, P. Shi, Q. Zhang, Fluorinated solid-electrolyte interphase in high-voltage lithium metal batteries. Joule 3(11), 2647–2661 (2019). https://doi.org/10.1016/j.joule.2019.09.022
L. Suo, O. Borodin, W. Sun, X. Fan, C. Yang et al., Advanced high-voltage aqueous lithium-ion battery enabled by “water-in-bisalt” electrolyte. Angew. Chem. Int. Ed. 55(25), 7136–7141 (2016). https://doi.org/10.1002/anie.201602397
T. Jin, X. Ji, P.F. Wang, K. Zhu, J. Zhang et al., High‐energy aqueous sodium‐ion batteries. Angew. Chem. Int. Ed. 60(21), 11943–11948 (2021). https://doi.org/10.1002/anie.202017167
L. Jiang, Y. Lu, C. Zhao, L. Liu, J. Zhang et al., Building aqueous K-ion batteries for energy storage. Nat. Energy 4, 495–503 (2019). https://doi.org/10.1038/s41560-019-0388-0
J. Wang, Y. Yamada, K. Sodeyama, C.H. Chiang, Y. Tateyama et al., Superconcentrated electrolytes for a high-voltage lithium-ion battery. Nat. Commun. 7, 12032 (2016). https://doi.org/10.1038/ncomms12032
L. Suo, W. Xue, M. Gobet, S.G. Greenbaum, C. Wang et al., Fluorine-donating electrolytes enable highly reversible 5-V-class Li metal batteries. Proc. Natl. Acad. Sci. U. S. A. 115(6), 1156–1161 (2018). https://doi.org/10.1073/pnas.1712895115
L. Xing, X. Zheng, M. Schroeder, J. Alvarado, A. von Wald Cresce et al., Deciphering the ethylene carbonate–propylene carbonate mystery in Li-ion batteries. Acc. Chem. Res. 51(2), 282–289 (2018). https://doi.org/10.1021/acs.accounts.7b00474
S.-K. Jeong, M. Inaba, Y. Iriyama, T. Abe, Z. Ogumi, Electrochemical intercalation of lithium ion within graphite from propylene carbonate solutions. Electrochem. Solid-State Lett. 6(1), A13 (2003). https://doi.org/10.1149/1.1526781
T. Doi, R. Masuhara, M. Hashinokuchi, Y. Shimizu, M. Inaba, Concentrated LiPF6/PC electrolyte solutions for 5-V LiNi0.5Mn1.5O4 positive electrode in lithium-ion batteries. Electrochim. Acta 209, 219–224 (2016). https://doi.org/10.1016/j.electacta.2016.05.062
X. Li, X. Ou, Y. Tang, 6.0 V high-voltage and concentrated electrolyte toward high energy density K-based dual-graphite battery. Adv. Energy Mater. 10(41), 2002567 (2020). https://doi.org/10.1002/aenm.202002567
K. Sodeyama, Y. Yamada, K. Aikawa, A. Yamada, Y. Tateyama, Sacrificial anion reduction mechanism for electrochemical stability improvement in highly concentrated Li-salt electrolyte. J. Phys. Chem. C 118(26), 14091–14097 (2014). https://doi.org/10.1021/jp501178n
X. Fan, L. Chen, X. Ji, T. Deng, S. Hou et al., Highly fluorinated interphases enable high-voltage Li-metal batteries. Chem 4(1), 174–185 (2018). https://doi.org/10.1016/j.chempr.2017.10.017
S. Liu, J. Mao, L. Zhang, W.K. Pang, A. Du et al., Manipulating the solvation structure of nonflammable electrolyte and interface to enable unprecedented stability of graphite anodes beyond 2 years for safe potassium-ion batteries. Adv. Mater. 33, 2006313 (2021). https://doi.org/10.1002/adma.202006313
Y. Yamada, K. Usui, C.H. Chiang, K. Kikuchi, K. Furukawa et al., General observation of lithium intercalation into graphite in ethylene-carbonate-free superconcentrated electrolytes. ACS Appl. Mater. Interfaces 6(14), 10892–10899 (2014). https://doi.org/10.1021/am5001163
Y. Yamada, Developing new functionalities of superconcentrated electrolytes for lithium-ion batteries. Electrochemistry 85(9), 559–565 (2017). https://doi.org/10.5796/electrochemistry.85.559
W. Liu, J. Li, W. Li, H. Xu, C. Zhang et al., Inhibition of transition metals dissolution in cobalt-free cathode with ultrathin robust interphase in concentrated electrolyte. Nat. Commun. 11, 3629 (2020). https://doi.org/10.1038/s41467-020-17396-x
S. Jiao, X. Ren, R. Cao, M.H. Engelhard, Y. Liu et al., Stable cycling of high-voltage lithium metal batteries in ether electrolytes. Nat. Energy 3(9), 739–746 (2018). https://doi.org/10.1038/s41560-018-0199-8
X. Ren, L. Zou, S. Jiao, D. Mei, M.H. Engelhard et al., High-concentration ether electrolytes for stable high-voltage lithium metal batteries. ACS Energy Lett. 4(4), 896–902 (2019). https://doi.org/10.1021/acsenergylett.9b00381
S.-K. Jeong, H.-Y. Seo, D.-H. Kim, H.-K. Han, J.-G. Kim et al., Suppression of dendritic lithium formation by using concentrated electrolyte solutions. Electrochem. Commun. 10(4), 635–638 (2008). https://doi.org/10.1016/j.elecom.2008.02.006
S. Wang, Y. Ding, G. Zhou, G. Yu, A. Manthiram, Durability of the Li1+xTi2–xAlx(PO4)3 solid electrolyte in lithium–sulfur batteries. ACS Energy Lett. 1(6), 1080–1085 (2016). https://doi.org/10.1021/acsenergylett.6b00481
L. Wang, Y. Qu, C. Su, R. Li, D. Liu et al., Interfacial engineering of Li anode and Ni-rich cathode via anion-regulated SEI/CEI layers in flame-retardant nanofiber-reinforced polymer electrolyte for lithium metal batteries. Adv. Energy Mater. 15(35), 2501185 (2025). https://doi.org/10.1002/aenm.202501185
Z. Wang, H. Tu, X. Ma, S. Lu, G. Su et al., Flame-retardant electrolytes with electrochemically-inert and weakly coordinating dichloroalkane diluents for practical lithium metal batteries. Nat. Commun. 16, 10188 (2025). https://doi.org/10.1038/s41467-025-65138-8
K. Lee, S.-H. Kwon, J. Kim, E. Park, I. Kim et al., Fluorinated cyclic ether diluent for high-voltage lithium metal batteries. ACS Energy Lett. 9(5), 2201–2211 (2024). https://doi.org/10.1021/acsenergylett.4c00481
X. Wang, Y. Bai, Z. Lv, Y. Liu, W. Dong et al., In situ polymerized localized high-concentration electrolytes for ultrahigh-rate sodium metal batteries. Adv. Energy Mater. 15(28), 2500887 (2025). https://doi.org/10.1002/aenm.202500887
D. Guo, J. Wang, Z. Cui, Z. Shi, G. Henkelman et al., Low-temperature sodium–sulfur batteries enabled by ionic liquid in localized high concentration electrolytes. Adv. Funct. Mater. 34(51), 2409494 (2024). https://doi.org/10.1002/adfm.202409494
Q. Wu, Y. Qi, Revealing heterogeneous electric double layer (EDL) structures of localized high-concentration electrolytes (LHCEs) and their impact on solid–electrolyte interphase (SEI) formation in lithium batteries. Energy Environ. Sci. 18(6), 3036–3046 (2025). https://doi.org/10.1039/d5ee00206k
Y. Chen, Z. Yu, P. Rudnicki, H. Gong, Z. Huang et al., Steric effect tuned ion solvation enabling stable cycling of high-voltage lithium metal battery. J. Am. Chem. Soc. 143(44), 18703–18713 (2021). https://doi.org/10.1021/jacs.1c09006
J. Choi, H. Jeong, J. Jang, A.-R. Jeon, I. Kang et al., Weakly solvating solution enables chemical prelithiation of graphite–SiOx anodes for high-energy Li-ion batteries. J. Am. Chem. Soc. 143(24), 9169–9176 (2021). https://doi.org/10.1021/jacs.1c03648
J. Holoubek, H. Liu, Z. Wu, Y. Yin, X. Xing et al., Tailoring electrolyte solvation for Li metal batteries cycled at ultra-low temperature. Nat. Energy 6(3), 303–313 (2021). https://doi.org/10.1038/s41560-021-00783-z
Y.-X. Yao, X. Chen, C. Yan, X.-Q. Zhang, W.-L. Cai et al., Regulating interfacial chemistry in lithium-ion batteries by a weakly solvating electrolyte. Angew. Chem. Int. Ed. 60(8), 4090–4097 (2021). https://doi.org/10.1002/anie.202011482
M. Mao, X. Ji, Q. Wang, Z. Lin, M. Li et al., Anion-enrichment interface enables high-voltage anode-free lithium metal batteries. Nat. Commun. 14, 1082 (2023). https://doi.org/10.1038/s41467-023-36853-x
X. Zheng, Z. Gu, J. Fu, H. Wang, X. Ye et al., Knocking down the kinetic barriers towards fast-charging and low-temperature sodium metal batteries. Energy Environ. Sci. 14(9), 4936–4947 (2021). https://doi.org/10.1039/d1ee01404h
S. Zhu, J. Chen, Dual strategy with Li-ion solvation and solid electrolyte interphase for high Coulombic efficiency of lithium metal anode. Energy Storage Mater. 44, 48–56 (2022). https://doi.org/10.1016/j.ensm.2021.10.007
H.M. Law, Z. Wang, S. Xu, L. Shen, B. Py et al., A weakly solvating solvent-based quasi-solid electrolyte for sodium metal batteries. Energy Environ. Sci. 18(19), 8838–8848 (2025). https://doi.org/10.1039/d5ee02153g
T. Jin, X.-Y. Li, M. Zhao, S. Feng, Z. Li et al., Promoting the rate performances of weakly solvating electrolyte-based lithium‒sulfur batteries. Angew. Chem. Int. Ed. 64(32), e202504898 (2025). https://doi.org/10.1002/anie.202504898
Z.-X. Chen, J.-J. Zhao, G.-Y. Fang, S. Zhang, J. Ma et al., Accelerating the sulfur redox kinetics in weakly-solvating lithium‒sulfur batteries under lean-electrolyte conditions. Energy Storage Mater. 83, 104502 (2025). https://doi.org/10.1016/j.ensm.2025.104502
D. Feng, Y. Xie, Y. Jiao, P. Wu, Leveraging cation effect for low temperature aqueous Zn-based batteries. Nat. Commun. 16, 9254 (2025). https://doi.org/10.1038/s41467-025-64278-1
N. Chang, T. Li, R. Li, S. Wang, Y. Yin et al., An aqueous hybrid electrolyte for low-temperature zinc-based energy storage devices. Energy Environ. Sci. 13(10), 3527–3535 (2020). https://doi.org/10.1039/d0ee01538e
J. Liu, C. Yang, X. Chi, B. Wen, W. Wang et al., Water/sulfolane hybrid electrolyte achieves ultralow-temperature operation for high-voltage aqueous lithium-ion batteries. Adv. Funct. Mater. 32, 2106811 (2022). https://doi.org/10.1002/adfm.202106811
P. Wang, T.C. Li, Y. Liu, C. Lin, Y. Cui et al., Targeted docking of localized hydrogen bond for efficient and reversible zinc-ion batteries. Angew. Chem. Int. Ed. 64(15), e202422547 (2025). https://doi.org/10.1002/anie.202422547
J. Hao, L. Yuan, C. Ye, D. Chao, K. Davey et al., Boosting zinc electrode reversibility in aqueous electrolytes by using low-cost antisolvents. Angew. Chem. Int. Ed. 60(13), 7366–7375 (2021). https://doi.org/10.1002/anie.202016531
Q. Zhang, Y. Ma, Y. Lu, L. Li, F. Wan et al., Modulating electrolyte structure for ultralow temperature aqueous zinc batteries. Nat. Commun. 11, 4463 (2020). https://doi.org/10.1038/s41467-020-18284-0
L. Cao, D. Li, F.A. Soto, V. Ponce, B. Zhang et al., Highly reversible aqueous zinc batteries enabled by zincophilic–zincophobic interfacial layers and interrupted hydrogen-bond electrolytes. Angew. Chem. Int. Ed. 60(34), 18845–18851 (2021). https://doi.org/10.1002/anie.202107378
A. von Wald Cresce, M. Gobet, O. Borodin, J. Peng, S.M. Russell et al., Anion solvation in carbonate-based electrolytes. J. Phys. Chem. C 119(49), 27255–27264 (2015). https://doi.org/10.1021/acs.jpcc.5b08895
K.W. Schroder, A.G. Dylla, L.D.C. Bishop, E.R. Kamilar, J. Saunders et al., Effects of solute–solvent hydrogen bonding on nonaqueous electrolyte structure. J. Phys. Chem. Lett. 6(15), 2888–2891 (2015). https://doi.org/10.1021/acs.jpclett.5b01216
X. Zhang, X. Dong, X. Yue, J. Gao, Z. Shi et al., Solvation regulation via hydrogen bonding to mitigate Al current collector corrosion for high-voltage Li-ion batteries. Adv. Energy Mater. 15(10), 2403588 (2025). https://doi.org/10.1002/aenm.202403588
Z. Yang, J. He, W.-H. Lai, J. Peng, X.-H. Liu et al., Fire-retardant, stable-cycling and high-safety sodium ion battery. Angew. Chem. Int. Ed. 60(52), 27086–27094 (2021). https://doi.org/10.1002/anie.202112382
Z. Yang, J. He, W.H. Lai, J. Peng, X.H. Liu et al., Fire-retardant, Stable-cycling and high-safety sodium ion battery. Angew. Chem. Int. Ed. 133(52), 27292–27300 (2021). https://doi.org/10.1002/anie.202112382
J.A.P. Coutinho, S.P. Pinho, Special issue on deep eutectic solvents: a foreword. Fluid Phase Equilib. 448, 1 (2017). https://doi.org/10.1016/j.fluid.2017.06.011
L. Geng, X. Wang, K. Han, P. Hu, L. Zhou et al., Eutectic electrolytes in advanced metal-ion batteries. ACS Energy Lett. 7(1), 247–260 (2022). https://doi.org/10.1021/acsenergylett.1c02088
C. Zhang, L. Zhang, G. Yu, Eutectic electrolytes as a promising platform for next-generation electrochemical energy storage. Acc. Chem. Res. 53(8), 1648–1659 (2020). https://doi.org/10.1021/acs.accounts.0c00360
X. Lu, E.J. Hansen, G. He, J. Liu, Eutectic electrolytes chemistry for rechargeable Zn batteries. Small 18(21), 2200550 (2022). https://doi.org/10.1002/smll.202200550
J. Wu, Q. Liang, X. Yu, Q.-F. Lü, L. Ma et al., Deep eutectic solvents for boosting electrochemical energy storage and conversion: a review and perspective. Adv. Funct. Mater. 31(22), 2011102 (2021). https://doi.org/10.1002/adfm.202011102
B.B. Hansen, S. Spittle, B. Chen, D. Poe, Y. Zhang et al., Deep eutectic solvents: a review of fundamentals and applications. Chem. Rev. 121(3), 1232–1285 (2021). https://doi.org/10.1021/acs.chemrev.0c00385
M.E. Di Pietro, A. Mele, Deep eutectics and analogues as electrolytes in batteries. J. Mol. Liq. 338, 116597 (2021). https://doi.org/10.1016/j.molliq.2021.116597
E.L. Smith, A.P. Abbott, K.S. Ryder, Deep eutectic solvents (DESs) and their applications. Chem. Rev. 114(21), 11060–11082 (2014). https://doi.org/10.1021/cr300162p
M. Fang, X. Yue, Y. Dong, Y. Chen, Z. Liang, A temperature-dependent solvating electrolyte for wide-temperature and fast-charging lithium metal batteries. Joule 8(1), 91–103 (2024). https://doi.org/10.1016/j.joule.2023.12.012
J. Zhao, J. Zhang, W. Yang, B. Chen, Z. Zhao et al., Water-in-deep eutectic solvent” electrolytes enable zinc metal anodes for rechargeable aqueous batteries. Nano Energy 57, 625–634 (2019). https://doi.org/10.1016/j.nanoen.2018.12.086
W. Yang, X. Du, J. Zhao, Z. Chen, J. Li et al., Hydrated eutectic electrolytes with ligand-oriented solvation shells for long-cycling zinc-organic batteries. Joule 4(7), 1557–1574 (2020). https://doi.org/10.1016/j.joule.2020.05.018
Y. Hu, Novel room temperature molten salt electrolyte based on LiTFSI and acetamide for lithium batteries. Electrochem. Commun. 6(1), 28–32 (2004). https://doi.org/10.1016/j.elecom.2003.10.009
A. Boisset, J. Jacquemin, M. Anouti, Physical properties of a new deep eutectic solvent based on lithium bis [(trifluoromethyl)sulfonyl] imide and N-methylacetamide as superionic suitable electrolyte for lithium ion batteries and electric double layer capacitors. Electrochim. Acta 102, 120–126 (2013). https://doi.org/10.1016/j.electacta.2013.03.150
A. Boisset, S. Menne, J. Jacquemin, A. Balducci, M. Anouti, Deep eutectic solvents based on N-methylacetamide and a lithium salt as suitable electrolytes for lithium-ion batteries. Phys. Chem. Chem. Phys. 15(46), 20054–20063 (2013). https://doi.org/10.1039/c3cp53406e
Z. Hu, F. Xian, Z. Guo, C. Lu, X. Du et al., Nonflammable nitrile deep eutectic electrolyte enables high-voltage lithium metal batteries. Chem. Mater. 32(8), 3405–3413 (2020). https://doi.org/10.1021/acs.chemmater.9b05003
Y. Lu, Z. Tu, L.A. Archer, Stable lithium electrodeposition in liquid and nanoporous solid electrolytes. Nat. Mater. 13(10), 961–969 (2014). https://doi.org/10.1038/nmat4041
J. Qian, W.A. Henderson, W. Xu, P. Bhattacharya, M. Engelhard et al., High rate and stable cycling of lithium metal anode. Nat. Commun. 6, 6362 (2015). https://doi.org/10.1038/ncomms7362
C. Guo, K. Zhang, Q. Zhao, L. Pei, J. Chen, High-performance sodium batteries with the 9, 10-anthraquinone/CMK-3 cathode and an ether-based electrolyte. Chem. Commun. 51(50), 10244–10247 (2015). https://doi.org/10.1039/c5cc02251g
R. Cao, K. Mishra, X. Li, J. Qian, M.H. Engelhard et al., Enabling room temperature sodium metal batteries. Nano Energy 30, 825–830 (2016). https://doi.org/10.1016/j.nanoen.2016.09.013
L. Schafzahl, I. Hanzu, M. Wilkening, S.A. Freunberger, An electrolyte for reversible cycling of sodium metal and intercalation compounds. Chemsuschem 10(2), 401–408 (2017). https://doi.org/10.1002/cssc.201601222
I.A. Shkrob, T.W. Marin, Y. Zhu, D.P. Abraham, Why bis(fluorosulfonyl)imide is a “magic anion” for electrochemistry. J. Phys. Chem. C 118(34), 19661–19671 (2014). https://doi.org/10.1021/jp506567p
J. Lee, Y. Lee, J. Lee, S.-M. Lee, J.-H. Choi et al., Ultraconcentrated sodium bis(fluorosulfonyl)imide-based electrolytes for high-performance sodium metal batteries. ACS Appl. Mater. Interfaces 9(4), 3723–3732 (2017). https://doi.org/10.1021/acsami.6b14878
M. He, K.C. Lau, X. Ren, N. Xiao, W.D. McCulloch et al., Concentrated electrolyte for the sodium–oxygen battery: solvation structure and improved cycle life. Angew. Chem. Int. Ed. 55(49), 15310–15314 (2016). https://doi.org/10.1002/anie.201608607
X. Shi, J. Xie, J. Wang, S. Xie, Z. Yang et al., A weakly solvating electrolyte towards practical rechargeable aqueous zinc-ion batteries. Nat. Commun. 15, 302 (2024). https://doi.org/10.1038/s41467-023-44615-y
T. Li, H. Yang, X. Dong, H. Ma, J. Cai et al., Co-regulation of interface and bulk for enhanced localized high-concentration electrolytes in stable and practical zinc metal batteries. Angew. Chem. Int. Ed. 64(29), e202501183 (2025). https://doi.org/10.1002/anie.202501183
C. Huang, M. Zhao, C. Xu, Y. Lv, M. Fang et al., Engineering anion-diluent matrix for ion-decoupled localized high-concentration electrolytes toward highly stable aqueous zinc ion batteries. Angew. Chem. Int. Ed. 64(40), e202511410 (2025). https://doi.org/10.1002/anie.202511410
Y. Yamada, Y. Takazawa, K. Miyazaki, T. Abe, Electrochemical lithium intercalation into graphite in dimethyl sulfoxide-based electrolytes: effect of solvation structure of lithium ion. J. Phys. Chem. C 114(26), 11680–11685 (2010). https://doi.org/10.1021/jp1037427
F. Li, L. Kong, Y. Sun, Y. Jin, P. Hou, Micron-sized monocrystalline LiNi1/3Co1/3Mn1/3O2 as high-volumetric-energy-density cathode for lithium-ion batteries. J. Mater. Chem. A 6, 12344–12352 (2018). https://doi.org/10.1039/c8ta03363c
A. Manthiram, A reflection on lithium-ion battery cathode chemistry. Nat. Commun. 11, 1550 (2020). https://doi.org/10.1038/s41467-020-15355-0
B. Wang, H.A. Doan, S.-B. Son, D.P. Abraham, S.E. Trask et al., Data-driven design of electrolyte additives supporting high-performance 5 V LiNi0.5Mn1.5O4 positive electrodes. Nat. Commun. 16, 3413 (2025). https://doi.org/10.1038/s41467-025-57961-w
Z. Zhang, M. Avdeev, H. Chen, W. Yin, W.H. Kan et al., Lithiated Prussian blue analogues as positive electrode active materials for stable non-aqueous lithium-ion batteries. Nat. Commun. 13, 7790 (2022). https://doi.org/10.1038/s41467-022-35376-1
C.S. Johnson, J.-S. Kim, C. Lefief, N. Li, J.T. Vaughey et al., The significance of the Li2MnO3 component in ‘composite’ xLi2MnO3·(1–x)LiMn0.5Ni0.5O2 electrodes. Electrochem. Commun. 6(10), 1085–1091 (2004). https://doi.org/10.1016/j.elecom.2004.08.002
D. Aurbach, B.D. McCloskey, L.F. Nazar, P.G. Bruce, Advances in understanding mechanisms underpinning lithium–air batteries. Nat. Energy 1(9), 16128 (2016). https://doi.org/10.1038/nenergy.2016.128
P. Hartmann, C.L. Bender, M. Vračar, A.K. Dürr, A. Garsuch et al., A rechargeable room-temperature sodium superoxide (NaO2) battery. Nat. Mater. 12(3), 228–232 (2013). https://doi.org/10.1038/nmat3486
Z. Peng, S.A. Freunberger, L.J. Hardwick, Y. Chen, V. Giordani et al., Oxygen reactions in a non-aqueous Li+ electrolyte. Angew. Chem. Int. Ed. 50(28), 6351–6355 (2011). https://doi.org/10.1002/anie.201100879
Y.-C. Lu, B.M. Gallant, D.G. Kwabi, J.R. Harding, R.R. Mitchell et al., Lithium–oxygen batteries: bridging mechanistic understanding and battery performance. Energy Environ. Sci. 6(3), 750 (2013). https://doi.org/10.1039/c3ee23966g
Z.-L. Wang, D. Xu, J.-J. Xu, X.-B. Zhang, Oxygen electrocatalysts in metal–air batteries: from aqueous to nonaqueous electrolytes. Chem. Soc. Rev. 43(22), 7746–7786 (2014). https://doi.org/10.1039/c3cs60248f
S.A. Freunberger, Y. Chen, Z. Peng, J.M. Griffin, L.J. Hardwick et al., Reactions in the rechargeable lithium–O2 battery with alkyl carbonate electrolytes. J. Am. Chem. Soc. 133(20), 8040–8047 (2011). https://doi.org/10.1021/ja2021747
C. Laoire, S. Mukerjee, E.J. Plichta, M.A. Hendrickson, K. Abraham, Rechargeable lithium/TEGDME-LiPF6/O2 battery. J. Electrochem. Soc. 158(3), A302–A308 (2011). https://doi.org/10.1149/1.3531981
R.S. Assary, J. Lu, P. Du, X. Luo, X. Zhang et al., The effect of oxygen crossover on the anode of a Li–O2 battery using an ether-based solvent: insights from experimental and computational studies. ChemSusChem 6(1), 51–55 (2013). https://doi.org/10.1002/cssc.201200810
C.O. Laoire, S. Mukerjee, K.M. Abraham, E.J. Plichta, M.A. Hendrickson, Influence of nonaqueous solvents on the electrochemistry of oxygen in the rechargeable lithium–air battery. J. Phys. Chem. C 114(19), 9178–9186 (2010). https://doi.org/10.1021/jp102019y
D. Xu, Z.-L. Wang, J.-J. Xu, L.-L. Zhang, L.-M. Wang et al., A stable sulfone based electrolyte for high performance rechargeable Li–O2 batteries. Chem. Commun. 48(95), 11674 (2012). https://doi.org/10.1039/c2cc36815c
B. Sun, C. Pompe, S. Dongmo, J. Zhang, K. Kretschmer et al., Challenges for developing rechargeable room-temperature sodium oxygen batteries. Adv. Mater. Technol. 3(9), 1800110 (2018). https://doi.org/10.1002/admt.201800110
Y. Wang, Y. Wang, Y.-X. Wang, X. Feng, W. Chen et al., Developments and perspectives on emerging high-energy-density sodium-metal batteries. Chem 5(10), 2547–2570 (2019). https://doi.org/10.1016/j.chempr.2019.05.026
J. Lu, Y. Jung Lee, X. Luo, K. Chun Lau, M. Asadi et al., A lithium–oxygen battery based on lithium superoxide. Nature 529(7586), 377–382 (2016). https://doi.org/10.1038/nature16484
W.A. Henderson, F. McKenna, M.A. Khan, N.R. Brooks, V.G. Young Jr. et al., Glyme–Lithium Bis(trifluoromethanesulfonyl)imide and Glyme–Lithium Bis(perfluoroethanesulfonyl)imide phase behavior and solvate structures. Chem. Mater. 17(9), 2284–2289 (2005). https://doi.org/10.1021/cm047881j
Y. Liu, L. Suo, H. Lin, W. Yang, Y. Fang et al., Novel approach for a high-energy-density Li–air battery: tri-dimensional growth of Li2O2 crystals tailored by electrolyte Li+ ion concentrations. J. Mater. Chem. A 2(24), 9020–9024 (2014). https://doi.org/10.1039/c4ta00834k
B. Liu, W. Xu, P. Yan, X. Sun, M.E. Bowden et al., Enhanced cycling stability of rechargeable Li–O2 batteries using high-concentration electrolytes. Adv. Funct. Mater. 26(4), 605–613 (2016). https://doi.org/10.1002/adfm.201503697
N. Tachikawa, K. Yamauchi, E. Takashima, J.-W. Park, K. Dokko et al., Reversibility of electrochemical reactions of sulfur supported on inverse opal carbon in glyme–Li salt molten complex electrolytes. Chem. Commun. 47(28), 8157–8159 (2011). https://doi.org/10.1039/c1cc12415c
X.-Y. Li, M. Zhao, Y.-W. Song, C.-X. Bi, Z. Li et al., Polysulfide chemistry in metal–sulfur batteries. Chem. Soc. Rev. 54(10), 4822–4873 (2025). https://doi.org/10.1039/d4cs00318g
L. Suo, Y.-S. Hu, H. Li, M. Armand, L. Chen, A new class of solvent-in-salt electrolyte for high-energy rechargeable metallic lithium batteries. Nat. Commun. 4, 1481 (2013). https://doi.org/10.1038/ncomms2513
K. Dokko, N. Tachikawa, K. Yamauchi, M. Tsuchiya, A. Yamazaki et al., Solvate ionic liquid electrolyte for Li–S batteries. J. Electrochem. Soc. 160(8), A1304–A1310 (2013). https://doi.org/10.1149/2.111308jes
M. Cuisinier, P.-E. Cabelguen, B.D. Adams, A. Garsuch, M. Balasubramanian et al., Unique behaviour of nonsolvents for polysulphides in lithium–sulphur batteries. Energy Environ. Sci. 7(8), 2697–2705 (2014). https://doi.org/10.1039/c4ee00372a
J. Zheng, X. Fan, G. Ji, H. Wang, S. Hou et al., Manipulating electrolyte and solid electrolyte interphase to enable safe and efficient Li–S batteries. Nano Energy 50, 431–440 (2018). https://doi.org/10.1016/j.nanoen.2018.05.065
X.-Y. Li, B.-Q. Li, S. Feng, Z. Li, L. Shen et al., Two-stage solvation of lithium polysulfides in working lithium–sulfur batteries. J. Am. Chem. Soc. 147(18), 15435–15447 (2025). https://doi.org/10.1021/jacs.5c01588
Q. Pang, X. Liang, C.Y. Kwok, L.F. Nazar, Advances in lithium–sulfur batteries based on multifunctional cathodes and electrolytes. Nat. Energy 1(9), 16132 (2016). https://doi.org/10.1038/nenergy.2016.132
Y. Yamada, M. Yaegashi, T. Abe, A. Yamada, A superconcentrated ether electrolyte for fast-charging Li-ion batteries. Chem. Commun. 49(95), 11194 (2013). https://doi.org/10.1039/c3cc46665e
K. Yoshida, M. Nakamura, Y. Kazue, N. Tachikawa, S. Tsuzuki et al., Oxidative-stability enhancement and charge transport mechanism in glyme–lithium salt equimolar complexes. J. Am. Chem. Soc. 133(33), 13121–13129 (2011). https://doi.org/10.1021/ja203983r
F. Li, T. Zhang, Y. Yamada, A. Yamada, H. Zhou, Enhanced cycling performance of Li-O2 batteries by the optimized electrolyte concentration of LiTFSA in glymes. Adv. Energy Mater. 3(4), 532–538 (2013). https://doi.org/10.1002/aenm.201200776
K. Matsumoto, K. Inoue, K. Nakahara, R. Yuge, T. Noguchi et al., Suppression of aluminum corrosion by using high concentration LiTFSI electrolyte. J. Power. Sources 231, 234–238 (2013). https://doi.org/10.1016/j.jpowsour.2012.12.028
L. Suo, Y.-S. Hu, H. Li, M. Armand, L. Chen, A new class of solvent-in-salt electrolyte for high-energy rechargeable metallic lithium batteries. Nat. Commun. 4, 1481 (2013). https://doi.org/10.1038/ncomms2513
S.-I. Okuoka, Y. Ogasawara, Y. Suga, M. Hibino, T. Kudo et al., A new sealed lithium-peroxide battery with a Co-doped Li2O cathode in a superconcentrated lithium bis(fluorosulfonyl)amide electrolyte. Sci. Rep. 4, 5684 (2014). https://doi.org/10.1038/srep05684
C. Zhang, A. Yamazaki, J. Murai, J.-W. Park, T. Mandai et al., Chelate effects in glyme/lithium bis(trifluoromethanesulfonyl)amide solvate ionic liquids, part 2: importance of solvate-structure stability for electrolytes of lithium batteries. J. Phys. Chem. C 118(31), 17362–17373 (2014). https://doi.org/10.1021/jp504099q
H. Kim, F. Wu, J.T. Lee, N. Nitta, H.-T. Lin et al., In situ formation of protective coatings on sulfur cathodes in lithium batteries with LiFSI-based organic electrolytes. Adv. Energy Mater. 5(6), 1401792 (2015). https://doi.org/10.1002/aenm.201401792
R. Petibon, C.P. Aiken, L. Ma, D. Xiong, J.R. Dahn, The use of ethyl acetate as a sole solvent in highly concentrated electrolyte for Li-ion batteries. Electrochim. Acta 154, 287–293 (2015). https://doi.org/10.1016/j.electacta.2014.12.093
Y. Yamada, C.H. Chiang, K. Sodeyama, J. Wang, Y. Tateyama et al., Corrosion prevention mechanism of aluminum metal in superconcentrated electrolytes. ChemElectroChem 2(11), 1687–1694 (2015). https://doi.org/10.1002/celc.201500235
J. Alvarado, M.A. Schroeder, M. Zhang, O. Borodin, E. Gobrogge et al., A carbonate-free, sulfone-based electrolyte for high-voltage Li-ion batteries. Mater. Today 21(4), 341–353 (2018). https://doi.org/10.1016/j.mattod.2018.02.005
Z. Zeng, V. Murugesan, K.S. Han, X. Jiang, Y. Cao et al., Non-flammable electrolytes with high salt-to-solvent ratios for Li-ion and Li-metal batteries. Nat. Energy 3(8), 674–681 (2018). https://doi.org/10.1038/s41560-018-0196-y
Z. Wang, Y. Sun, Y. Mao, F. Zhang, L. Zheng et al., Highly concentrated dual-anion electrolyte for non-flammable high-voltage Li-metal batteries. Energy Storage Mater. 30, 228–237 (2020). https://doi.org/10.1016/j.ensm.2020.05.020
M. Zhao, G. Xu, D. Lu, B. Xie, L. Huang et al., Formulating a non-flammable highly concentrated dual-salt electrolyte for wide temperature high-nickel lithium ion batteries. J. Electrochem. Soc. 168(5), 050511 (2021). https://doi.org/10.1149/1945-7111/abfb39
W. Cai, Y. Deng, Z. Deng, Y. Jia, Z. Li et al., Quasi-localized high-concentration electrolytes for high-voltage lithium metal batteries. Adv. Energy Mater. 13(31), 2301396 (2023). https://doi.org/10.1002/aenm.202301396
F. Hai, Y. Yi, Z. Xiao, J. Guo, X. Gao et al., A low-cost, fluorine-free localized highly concentrated electrolyte toward ultra-high loading lithium metal batteries. Adv. Energy Mater. 14(17), 2304253 (2024). https://doi.org/10.1002/aenm.202304253
Z. Han, L. Chen, G. Zheng, D. Zhang, K. Yang et al., Ionic liquid reinforcing ether coordination of localized high concentration electrolyte enables high-voltage lithium metal batteries. Adv. Mater. 37(14), 2416668 (2025). https://doi.org/10.1002/adma.202416668
J. Peng, H. Zhang, Z. Zeng, H. Zhang, H. Pei et al., Tailoring solvation structures via precise diluent engineering for high-rate 500 wh kg–1 lithium-metal batteries. Adv. Mater. 37(45), e09109 (2025). https://doi.org/10.1002/adma.202509109
T. Zhang, J. Yu, T. Lin, Y. Song, M. Li et al., Fluorinated deep eutectic gel electrolytes for sustainable lithium metal batteries. J. Am. Chem. Soc. 147(36), 32861–32872 (2025). https://doi.org/10.1021/jacs.5c08642
C. Luo, C. Ning, X. Huang, P. Zhang, L. Yang et al., Molecular engineering of weakly solvating dinitrile electrolytes for long-lasting room-temperature lithium metal batteries. Angew. Chem. Int. Ed. 64(29), e202507051 (2025). https://doi.org/10.1002/anie.202507051
H. Yang, H. Li, J. Li, Z. Sun, K. He et al., The rechargeable aluminum battery: opportunities and challenges. Angew. Chem. Int. Ed. 58(35), 11978–11996 (2019). https://doi.org/10.1002/anie.201814031
E. Miele, W.M. Dose, I. Manyakin, M.H. Frosz, Z. Ruff et al., Hollow-core optical fibre sensors for operando Raman spectroscopy investigation of Li-ion battery liquid electrolytes. Nat. Commun. 13, 1651 (2022). https://doi.org/10.1038/s41467-022-29330-4
Y.-H. Wang, S. Zheng, W.-M. Yang, R.-Y. Zhou, Q.-F. He et al., In situ Raman spectroscopy reveals the structure and dissociation of interfacial water. Nature 600, 81–85 (2021). https://doi.org/10.1038/s41586-021-04068-z
Y. Yamada, J. Wang, S. Ko, E. Watanabe, A. Yamada, Advances and issues in developing salt-concentrated battery electrolytes. Nat. Energy 4(4), 269–280 (2019). https://doi.org/10.1038/s41560-019-0336-z
N. Yao, X. Chen, Z.-H. Fu, Q. Zhang, Applying classical, Ab initio, and machine-learning molecular dynamics simulations to the liquid electrolyte for rechargeable batteries. Chem. Rev. 122(12), 10970–11021 (2022). https://doi.org/10.1021/acs.chemrev.1c00904
H. Wang, L. Zhang, J. Han, E. Weinan, DeePMD-kit: a deep learning package for many-body potential energy representation and molecular dynamics. Comput. Phys. Commun. 228, 178–184 (2018). https://doi.org/10.1016/j.cpc.2018.03.016
D. Lu, H. Wang, M. Chen, L. Lin, R. Car et al., 86 PFLOPS deep potential molecular dynamics simulation of 100 million atoms with ab initio accuracy. Comput. Phys. Commun. 259, 107624 (2021). https://doi.org/10.1016/j.cpc.2020.107624
Y. Hu, H. Su, J. Fu, J. Luo, Q. Yu et al., Solid solvation structure design improves all-solid-state organic batteries. Nat. Chem. 17(9), 1313–1322 (2025). https://doi.org/10.1038/s41557-025-01866-0
Q. You, Y. Sun, F. Wang, J. Cheng, F. Tang, Decoding the competing effects of dynamic solvation structures on nuclear magnetic resonance chemical shifts of battery electrolytes via machine learning. J. Am. Chem. Soc. 147(17), 14667–14676 (2025). https://doi.org/10.1021/jacs.5c02710
S. Chen, G. Wu, H. Jiang, J. Wang, T. Chen et al., External Li supply reshapes Li deficiency and lifetime limit of batteries. Nature 638(8051), 676–683 (2025). https://doi.org/10.1038/s41586-024-08465-y