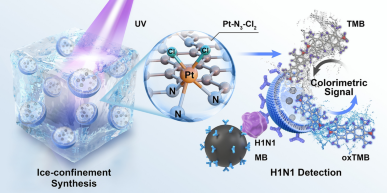
Confined Synthesis of Axial Chlorine Coordinated Single-Atom Nanozyme Within Liposomes for Sensitive Immunoassay
Corresponding Author: Wenhua Zhou
Nano-Micro Letters,
Vol. 18 (2026), Article Number: 293
Abstract
Single-atom nanozymes (SANs) are considered as the most promising candidates for signal amplification in biosensing applications. However, maintaining the integrity of active sites during functionalization while preserving catalytic performance in biological environments remains a critical challenge. Here, we present a low-temperature-photochemical synthesis strategy for constructing platinum single-atoms within liposomes (PtSANs@Lipo), achieving atomic-level regulation of SANs active sites through axial chlorine (Cl) coordination engineering. The PtN3Cl2 coordination structure, validated by synchrotron X-ray absorption spectroscopy, induces optimal d-band center modulation giving a remarkably low determining step energy barrier compared to conventional PtN3 configurations. This architecture further enables non-contact functionalization via liposomal encapsulation, fully preserving catalytic activity while preventing catalytic sites occupation by antibody, thereby reducing the Michaelis constant (Km) value by 50-fold compared to direct modification onto Pt single-atoms. The constructed immunosensor based on PtSANs@Lipo demonstrates highly sensitive detection of viral pathogens, including influenza A virus H1N1, SARS-CoV-2, and influenza B virus antigens with limits of detection as low as 0.42, 2.23, and 0.36 fg mL−1, respectively. This work establishes a paradigm for bio-adaptive nanozyme design through synergistic coordination engineering and liposomal functionalization architectures, and may thus provide a universal approach for signal amplification in point-of-care diagnostics.
Highlights:
1 Axial chlorine coordination engineering constructs PtN3Cl2 single-atom nanozymes inside liposomes via a low-temperature photochemical method.
2 The unique structure lowers the reaction energy barrier and enables antibody conjugation without blocking active sites, reducing Km by 50-fold.
3 The resulting immunosensor achieves ultra-sensitive detection of viral antigens with detection limits as low as 0.36 fg mL-1.
Keywords
Download Citation
Endnote/Zotero/Mendeley (RIS)BibTeX
- S. Ding, J.A. Barr, Z. Lyu, F. Zhang, M. Wang et al., Effect of phosphorus modulation in iron single-atom catalysts for peroxidase mimicking. Adv. Mater. 36(10), e2209633 (2024). https://doi.org/10.1002/adma.202209633
- E.M. Hamed, L. He, V. Rai, S. Hu, S.F.Y. Li, Copper single-atom nanozyme mimicking galactose oxidase with superior catalytic activity and selectivity. Small 20(49), e2405986 (2024). https://doi.org/10.1002/smll.202405986
- E.M. Hamed, F.M. Fung, S.F.Y. Li, Zinc single-atom nanozyme as carbonic anhydrase mimic for CO2 capture and conversion. ACS Mater. Au 5(2), 377–384 (2025). https://doi.org/10.1021/acsmaterialsau.4c00156
- T. Chen, H. Guo, X. Wen, J. Yu, H. Shi et al., Phosphatase-like nanozymes with tailed selectivity and enhanced activity for endotoxin sensing. Talanta 298, 128860 (2026). https://doi.org/10.1016/j.talanta.2025.128860
- B. Xu, Y. Cui, W. Wang, S. Li, C. Lyu et al., Immunomodulation-enhanced nanozyme-based tumor catalytic therapy. Adv. Mater. 32(33), e2003563 (2020). https://doi.org/10.1002/adma.202003563
- R. Wang, X. Ma, E.M. Hamed, B. Cao, L. Wang et al., Deciphering of laccase-like activity ruthenium single-atom nanozyme for identification/quantification and remediation of phenolic pollutants. Sens. Actuators B Chem. 426, 137112 (2025). https://doi.org/10.1016/j.snb.2024.137112
- E.M. Hamed, F.M. Fung, S.F.Y. Li, Bimetallic Cu/Zn single-atom nanozyme with superoxide dismutase-like activity. Small 21(36), e03879 (2025). https://doi.org/10.1002/smll.202503879
- Y. Ai, Z.-N. Hu, X. Liang, H.-B. Sun, H. Xin et al., Recent advances in nanozymes: from matters to bioapplications. Adv. Funct. Mater. 32(14), 2110432 (2022). https://doi.org/10.1002/adfm.202110432
- Z. Yu, Z. Xu, R. Zeng, M. Xu, H. Zheng et al., D-band-center-engineered platinum-based nanozyme for personalized pharmacovigilance. Angew. Chem. 137(2), e202414625 (2025). https://doi.org/10.1002/ange.202414625
- Z. Yu, Z. Xu, R. Zeng, M. Xu, H. Zheng et al., D-band-center-engineered platinum-based nanozyme for personalized pharmacovigilance. Angew. Chem. Int. Ed. Engl. 137(2), e202414625 (2025). https://doi.org/10.1002/ange.202414625
- R. Zeng, Q. Gao, L. Xiao, W. Wang, Y. Gu et al., Precise tuning of the D-band center of dual-atomic enzymes for catalytic therapy. J. Am. Chem. Soc. 146(14), 10023–10031 (2024). https://doi.org/10.1021/jacs.4c00791
- H. Gu, J. Li, P. Dai, T. Sun, C. Chen et al., Polyphenol oxidase-like nanozymes. Adv. Mater. 37(43), e09346 (2025). https://doi.org/10.1002/adma.202509346
- J. Li, X. Cai, P. Jiang, H. Wang, S. Zhang et al., Co-based nanozymatic profiling: advances spanning chemistry, biomedical, and environmental sciences. Adv. Mater. 36(8), e2307337 (2024). https://doi.org/10.1002/adma.202307337
- M. Tang, J. Ni, Z. Yue, T. Sun, C. Chen et al., Polyoxometalate-nanozyme-integrated nanomotors (POMotors) for self-propulsion-promoted synergistic photothermal-catalytic tumor therapy. Angew. Chem. Int. Ed. 63(6), e202315031 (2024). https://doi.org/10.1002/anie.202315031
- Z. Fu, K. Fan, X. He, Q. Wang, J. Yuan et al., Single-atom-based nanoenzyme in tissue repair. ACS Nano 18(20), 12639–12671 (2024). https://doi.org/10.1021/acsnano.4c00308
- S. Zhang, Y. Li, S. Sun, L. Liu, X. Mu et al., Single-atom nanozymes catalytically surpassing naturally occurring enzymes as sustained stitching for brain trauma. Nat. Commun. 13(1), 4744 (2022). https://doi.org/10.1038/s41467-022-32411-z
- Z. Guo, J. Hong, N. Song, M. Liang, Single-atom nanozymes: from precisely engineering to extensive applications. Acc. Mater. Res. 5(3), 347–357 (2024). https://doi.org/10.1021/accountsmr.3c00250
- C. Peng, R. Pang, J. Li, E. Wang, Current advances on the single-atom nanozyme and its bioapplications. Adv. Mater. 36(10), e2211724 (2024). https://doi.org/10.1002/adma.202211724
- E.M. Hamed, F.M. Fung, S.F.Y. Li, Unleashing the potential of single-atom nanozymes: catalysts for the future. ACS Sens. 9(8), 3840–3847 (2024). https://doi.org/10.1021/acssensors.4c00630
- E.M. Hamed, V. Rai, S.F.Y. Li, Single-atom nanozymes with peroxidase-like activity: a review. Chemosphere 346, 140557 (2024). https://doi.org/10.1016/j.chemosphere.2023.140557
- E.M. Hamed, M.M. Elsaady, S.F.Y. Li, Single-atom nanozymes in analytical chemistry: opportunities and challenges. Anal. Chem. 97(48), 26313–26325 (2025). https://doi.org/10.1021/acs.analchem.5c05006
- P. Muhammad, A. Zada, J. Rashid, S. Hanif, Y. Gao et al., Defect engineering in nanocatalysts: from design and synthesis to applications. Adv. Funct. Mater. 34(29), 2314686 (2024). https://doi.org/10.1002/adfm.202314686
- J. Shen, J. Chen, Y. Qian, X. Wang, D. Wang et al., Atomic engineering of single-atom nanozymes for biomedical applications. Adv. Mater. 36(21), e2313406 (2024). https://doi.org/10.1002/adma.202313406
- X. Zhang, G. Li, G. Chen, D. Wu, X. Zhou et al., Single-atom nanozymes: a rising star for biosensing and biomedicine. Coord. Chem. Rev. 418, 213376 (2020). https://doi.org/10.1016/j.ccr.2020.213376
- L. Jiao, W. Xu, Y. Wu, H. Yan, W. Gu et al., Single-atom catalysts boost signal amplification for biosensing. Chem. Soc. Rev. 50(2), 750–765 (2021). https://doi.org/10.1039/d0cs00367k
- S. Wei, M. Sun, J. Huang, Z. Chen, X. Wang et al., Axial chlorination engineering of single-atom nanozyme: Fe–N4Cl catalytic sites for efficient peroxidase-mimicking. J. Am. Chem. Soc. 146(48), 33239–33248 (2024). https://doi.org/10.1021/jacs.4c13335
- H.-Y. Lee, S.H.R. Shin, L.L. Abezgauz, S.A. Lewis, A.M. Chirsan et al., Integration of gold nanops into bilayer structures via adaptive surface chemistry. J. Am. Chem. Soc. 135(16), 5950–5953 (2013). https://doi.org/10.1021/ja400225n
- Y. Yu, Z. Zhu, H. Huang, Surface engineered single-atom systems for energy conversion. Adv. Mater. 36(16), e2311148 (2024). https://doi.org/10.1002/adma.202311148
- L. Zhang, M. Zhou, A. Wang, T. Zhang, Selective hydrogenation over supported metal catalysts: from nanops to single atoms. Chem. Rev. 120(2), 683–733 (2020). https://doi.org/10.1021/acs.chemrev.9b00230
- X.-F. Yang, A. Wang, B. Qiao, J. Li, J. Liu et al., Single-atom catalysts: a new frontier in heterogeneous catalysis. Acc. Chem. Res. 46(8), 1740–1748 (2013). https://doi.org/10.1021/ar300361m
- N. Kang, L. Liao, X. Zhang, Z. He, B. Yu et al., Engineering the axial coordination of cobalt single atom catalysts for efficient photocatalytic hydrogen evolution. Nano Res. 17(6), 5114–5121 (2024). https://doi.org/10.1007/s12274-024-6411-1
- J.-B. Pan, B.-H. Wang, S. Shen, L. Chen, S.-F. Yin, Introducing bidirectional axial coordination into BiVO4 @metal phthalocyanine core-shell photoanodes for efficient water oxidation. Angew. Chem. Int. Ed. Engl. 62(38), e202307246 (2023). https://doi.org/10.1002/anie.202307246
- D. Liu, X. Wan, J. Shui, Tailoring oxygen reduction reaction on M-N-C catalysts via axial coordination engineering. Small 20(50), e2406078 (2024). https://doi.org/10.1002/smll.202406078
- W. Song, C. Xiao, J. Ding, Z. Huang, X. Yang et al., Review of carbon support coordination environments for single metal atom electrocatalysts (SACS). Adv. Mater. 36(1), e2301477 (2024). https://doi.org/10.1002/adma.202301477
- L. Yin, M. Sun, S. Zhang, Y. Huang, B. Huang et al., Chlorine axial coordination activated lanthanum single atoms for efficient oxygen electroreduction with maximum utilization. Adv. Mater. 37(7), e2416387 (2025). https://doi.org/10.1002/adma.202416387
- S. Ren, Y. Wang, L. Shi, X. Xu, S. Zhong et al., Transforming plastics to single atom catalysts for peroxymonosulfate activation: axial chloride coordination intensified electron transfer pathway. Adv. Mater. 37(8), e2415339 (2025). https://doi.org/10.1002/adma.202415339
- Z. Zhang, Z. Feng, X. Zhao, D. Jean, Z. Yu et al., Functionalization and higher-order organization of liposomes with DNA nanostructures. Nat. Commun. 14(1), 5256 (2023). https://doi.org/10.1038/s41467-023-41013-2
- C. Wang, X. Lan, L. Zhu, Y. Wang, X. Gao et al., Construction strategy of functionalized liposomes and multidimensional application. Small 20(25), e2309031 (2024). https://doi.org/10.1002/smll.202309031
- V.P. Torchilin, Recent advances with liposomes as pharmaceutical carriers. Nat. Rev. Drug Discov. 4(2), 145–160 (2005). https://doi.org/10.1038/nrd1632
- N. Filipczak, J. Pan, S.S.K. Yalamarty, V.P. Torchilin, Recent advancements in liposome technology. Adv. Drug Deliv. Rev. 156, 4–22 (2020). https://doi.org/10.1016/j.addr.2020.06.022
- M. Lyu, M. Luo, J. Li, O.U. Akakuru, X. Fan et al., Personalized carbon monoxide-loaded biomimetic single-atom nanozyme for ferroptosis-enhanced FLASH radioimmunotherapy. Adv. Funct. Mater. 33(51), 2306930 (2023). https://doi.org/10.1002/adfm.202306930
- X.T. Le, N.T. Nguyen, W.T. Lee, Y. Yang, H.-G. Choi et al., Peroxidase-mimicking iron-based single-atom upconversion photocatalyst for enhancing chemodynamic therapy. Adv. Funct. Mater. 34(34), 2401893 (2024). https://doi.org/10.1002/adfm.202401893
- J.-H. Lee, Y. Shin, W. Lee, K. Whang, D. Kim et al., General and programmable synthesis of hybrid liposome/metal nanops. Sci. Adv. 2(12), e1601838 (2016). https://doi.org/10.1126/sciadv.1601838
- H. Zhang, G. Liu, L. Shi, J. Ye, Single-atom catalysts: emerging multifunctional materials in heterogeneous catalysis. Adv. Energy Mater. 8, 1701343 (2018). https://doi.org/10.1002/aenm.201701343
- Y. Zhang, J. Yang, R. Ge, J. Zhang, J.M. Cairney et al., The effect of coordination environment on the activity and selectivity of single-atom catalysts. Coord. Chem. Rev. 461, 214493 (2022). https://doi.org/10.1016/j.ccr.2022.214493
- J. Chang, W. Jing, X. Yong, A. Cao, J. Yu et al., Synthesis of ultrahigh-metal-density single-atom catalysts via metal sulfide-mediated atomic trapping. Nat. Synth 3(11), 1427–1438 (2024). https://doi.org/10.1038/s44160-024-00607-4
- D. Huang, L. Yin, X. Lu, S. Lin, Z. Niu et al., Directional electron transfer mechanisms with graphene quantum dots as the electron donor for photodecomposition of perfluorooctane sulfonate. Chem. Eng. J. 323, 406–414 (2017). https://doi.org/10.1016/j.cej.2017.04.124
- H. Wei, K. Huang, D. Wang, R. Zhang, B. Ge et al., Iced photochemical reduction to synthesize atomically dispersed metals by suppressing nanocrystal growth. Nat. Commun. 8(1), 1490 (2017). https://doi.org/10.1038/s41467-017-01521-4
- F. Maurer, J. Jelic, J. Wang, A. Gänzler, P. Dolcet et al., Tracking the formation, fate and consequence for catalytic activity of Pt single sites on CeO2. Nat. Catal. 3(10), 824–833 (2020). https://doi.org/10.1038/s41929-020-00508-7
- X. Liu, X. Song, G. Jiang, L. Tao, Z. Jin et al., Pt single-atom collaborate with Pt atom-clusters by an in-situ confined strategy for accelerating electrocatalytic hydrogen evolution. Chem. Eng. J. 481, 148430 (2024). https://doi.org/10.1016/j.cej.2023.148430
- Z. Xu, J. Jiang, Y. Li, T. Hu, J. Gu et al., Shape-regulated photothermal-catalytic tumor therapy using Polydopamine@Pt nanozymes with the elicitation of an immune response. Small 20(20), e2309096 (2024). https://doi.org/10.1002/smll.202309096
- K. Deng, H. Hu, Y. Li, X. Li, H. Deng et al., Mechanistic investigation and dual-mode colorimetric-chemiluminescent detection of glyphosate based on the specific inhibition of Fe3O4@Cu nanozyme peroxidase-like activity. Food Chem. 443, 138501 (2024). https://doi.org/10.1016/j.foodchem.2024.138501
- B.H.J. Hofstee, On the evaluation of the constants Vm and KM in enzyme reactions. Science 116(3013), 329–331 (1952). https://doi.org/10.1126/science.116.3013.329
- M. Sela, M. Poley, P. Mora-Raimundo, S. Kagan, A. Avital et al., Brain-targeted liposomes loaded with monoclonal antibodies reduce alpha-synuclein aggregation and improve behavioral symptoms in Parkinson’s disease. Adv. Mater. 35(51), e2304654 (2023). https://doi.org/10.1002/adma.202304654
- A.B. Ganganboina, A.D. Chowdhury, I.M. Khoris, F. Nasrin, K. Takemura et al., Dual modality sensor using liposome-based signal amplification technique for ultrasensitive norovirus detection. Biosens. Bioelectron. 157, 112169 (2020). https://doi.org/10.1016/j.bios.2020.112169
- G. Seo, G. Lee, M.J. Kim, S.-H. Baek, M. Choi et al., Rapid detection of COVID-19 causative virus (SARS-CoV-2) in human nasopharyngeal swab specimens using field-effect transistor-based biosensor. ACS Nano 14(4), 5135–5142 (2020). https://doi.org/10.1021/acsnano.0c02823
- J. Shang, Y. Wang, Y. Pang, M. Yu, Y. Wang et al., Point-of-care diagnosis of respiratory viruses at single-nucleotide resolution with an autocatalytic rolling circle amplification system. Angew. Chem. Int. Ed. 64(52), e18925 (2025). https://doi.org/10.1002/anie.202518925
- A. Piriya V S, P. Joseph, K. Daniel S C G, S. Lakshmanan, T. Kinoshita et al., Colorimetric sensors for rapid detection of various analytes. Mater. Sci. Eng. C Mater. Biol. Appl. 78, 1231–1245 (2017). https://doi.org/10.1016/j.msec.2017.05.018
- R. Cui, H. Tang, Q. Huang, T. Ye, J. Chen et al., AI-assisted smartphone-based colorimetric biosensor for visualized, rapid and sensitive detection of pathogenic bacteria. Biosens. Bioelectron. 259, 116369 (2024). https://doi.org/10.1016/j.bios.2024.116369
- S. Wang, Y. Zhu, Z. Zhou, Y. Luo, Y. Huang et al., Integrated ultrasound-enrichment and machine learning in colorimetric lateral flow assay for accurate and sensitive clinical Alzheimer’s biomarker diagnosis. Adv. Sci. 11(42), 2406196 (2024). https://doi.org/10.1002/advs.202406196
- T. He, Z. Zhang, H. Zhang, Z. Zhang, J. Xie et al., Bag of tricks for image classification with convolutional neural networks. 2019 IEEE/CVF Conference on Computer Vision and Pattern Recognition (CVPR)., 558–567. IEEE (2020). https://doi.org/10.1109/CVPR.2019.00065
References
S. Ding, J.A. Barr, Z. Lyu, F. Zhang, M. Wang et al., Effect of phosphorus modulation in iron single-atom catalysts for peroxidase mimicking. Adv. Mater. 36(10), e2209633 (2024). https://doi.org/10.1002/adma.202209633
E.M. Hamed, L. He, V. Rai, S. Hu, S.F.Y. Li, Copper single-atom nanozyme mimicking galactose oxidase with superior catalytic activity and selectivity. Small 20(49), e2405986 (2024). https://doi.org/10.1002/smll.202405986
E.M. Hamed, F.M. Fung, S.F.Y. Li, Zinc single-atom nanozyme as carbonic anhydrase mimic for CO2 capture and conversion. ACS Mater. Au 5(2), 377–384 (2025). https://doi.org/10.1021/acsmaterialsau.4c00156
T. Chen, H. Guo, X. Wen, J. Yu, H. Shi et al., Phosphatase-like nanozymes with tailed selectivity and enhanced activity for endotoxin sensing. Talanta 298, 128860 (2026). https://doi.org/10.1016/j.talanta.2025.128860
B. Xu, Y. Cui, W. Wang, S. Li, C. Lyu et al., Immunomodulation-enhanced nanozyme-based tumor catalytic therapy. Adv. Mater. 32(33), e2003563 (2020). https://doi.org/10.1002/adma.202003563
R. Wang, X. Ma, E.M. Hamed, B. Cao, L. Wang et al., Deciphering of laccase-like activity ruthenium single-atom nanozyme for identification/quantification and remediation of phenolic pollutants. Sens. Actuators B Chem. 426, 137112 (2025). https://doi.org/10.1016/j.snb.2024.137112
E.M. Hamed, F.M. Fung, S.F.Y. Li, Bimetallic Cu/Zn single-atom nanozyme with superoxide dismutase-like activity. Small 21(36), e03879 (2025). https://doi.org/10.1002/smll.202503879
Y. Ai, Z.-N. Hu, X. Liang, H.-B. Sun, H. Xin et al., Recent advances in nanozymes: from matters to bioapplications. Adv. Funct. Mater. 32(14), 2110432 (2022). https://doi.org/10.1002/adfm.202110432
Z. Yu, Z. Xu, R. Zeng, M. Xu, H. Zheng et al., D-band-center-engineered platinum-based nanozyme for personalized pharmacovigilance. Angew. Chem. 137(2), e202414625 (2025). https://doi.org/10.1002/ange.202414625
Z. Yu, Z. Xu, R. Zeng, M. Xu, H. Zheng et al., D-band-center-engineered platinum-based nanozyme for personalized pharmacovigilance. Angew. Chem. Int. Ed. Engl. 137(2), e202414625 (2025). https://doi.org/10.1002/ange.202414625
R. Zeng, Q. Gao, L. Xiao, W. Wang, Y. Gu et al., Precise tuning of the D-band center of dual-atomic enzymes for catalytic therapy. J. Am. Chem. Soc. 146(14), 10023–10031 (2024). https://doi.org/10.1021/jacs.4c00791
H. Gu, J. Li, P. Dai, T. Sun, C. Chen et al., Polyphenol oxidase-like nanozymes. Adv. Mater. 37(43), e09346 (2025). https://doi.org/10.1002/adma.202509346
J. Li, X. Cai, P. Jiang, H. Wang, S. Zhang et al., Co-based nanozymatic profiling: advances spanning chemistry, biomedical, and environmental sciences. Adv. Mater. 36(8), e2307337 (2024). https://doi.org/10.1002/adma.202307337
M. Tang, J. Ni, Z. Yue, T. Sun, C. Chen et al., Polyoxometalate-nanozyme-integrated nanomotors (POMotors) for self-propulsion-promoted synergistic photothermal-catalytic tumor therapy. Angew. Chem. Int. Ed. 63(6), e202315031 (2024). https://doi.org/10.1002/anie.202315031
Z. Fu, K. Fan, X. He, Q. Wang, J. Yuan et al., Single-atom-based nanoenzyme in tissue repair. ACS Nano 18(20), 12639–12671 (2024). https://doi.org/10.1021/acsnano.4c00308
S. Zhang, Y. Li, S. Sun, L. Liu, X. Mu et al., Single-atom nanozymes catalytically surpassing naturally occurring enzymes as sustained stitching for brain trauma. Nat. Commun. 13(1), 4744 (2022). https://doi.org/10.1038/s41467-022-32411-z
Z. Guo, J. Hong, N. Song, M. Liang, Single-atom nanozymes: from precisely engineering to extensive applications. Acc. Mater. Res. 5(3), 347–357 (2024). https://doi.org/10.1021/accountsmr.3c00250
C. Peng, R. Pang, J. Li, E. Wang, Current advances on the single-atom nanozyme and its bioapplications. Adv. Mater. 36(10), e2211724 (2024). https://doi.org/10.1002/adma.202211724
E.M. Hamed, F.M. Fung, S.F.Y. Li, Unleashing the potential of single-atom nanozymes: catalysts for the future. ACS Sens. 9(8), 3840–3847 (2024). https://doi.org/10.1021/acssensors.4c00630
E.M. Hamed, V. Rai, S.F.Y. Li, Single-atom nanozymes with peroxidase-like activity: a review. Chemosphere 346, 140557 (2024). https://doi.org/10.1016/j.chemosphere.2023.140557
E.M. Hamed, M.M. Elsaady, S.F.Y. Li, Single-atom nanozymes in analytical chemistry: opportunities and challenges. Anal. Chem. 97(48), 26313–26325 (2025). https://doi.org/10.1021/acs.analchem.5c05006
P. Muhammad, A. Zada, J. Rashid, S. Hanif, Y. Gao et al., Defect engineering in nanocatalysts: from design and synthesis to applications. Adv. Funct. Mater. 34(29), 2314686 (2024). https://doi.org/10.1002/adfm.202314686
J. Shen, J. Chen, Y. Qian, X. Wang, D. Wang et al., Atomic engineering of single-atom nanozymes for biomedical applications. Adv. Mater. 36(21), e2313406 (2024). https://doi.org/10.1002/adma.202313406
X. Zhang, G. Li, G. Chen, D. Wu, X. Zhou et al., Single-atom nanozymes: a rising star for biosensing and biomedicine. Coord. Chem. Rev. 418, 213376 (2020). https://doi.org/10.1016/j.ccr.2020.213376
L. Jiao, W. Xu, Y. Wu, H. Yan, W. Gu et al., Single-atom catalysts boost signal amplification for biosensing. Chem. Soc. Rev. 50(2), 750–765 (2021). https://doi.org/10.1039/d0cs00367k
S. Wei, M. Sun, J. Huang, Z. Chen, X. Wang et al., Axial chlorination engineering of single-atom nanozyme: Fe–N4Cl catalytic sites for efficient peroxidase-mimicking. J. Am. Chem. Soc. 146(48), 33239–33248 (2024). https://doi.org/10.1021/jacs.4c13335
H.-Y. Lee, S.H.R. Shin, L.L. Abezgauz, S.A. Lewis, A.M. Chirsan et al., Integration of gold nanops into bilayer structures via adaptive surface chemistry. J. Am. Chem. Soc. 135(16), 5950–5953 (2013). https://doi.org/10.1021/ja400225n
Y. Yu, Z. Zhu, H. Huang, Surface engineered single-atom systems for energy conversion. Adv. Mater. 36(16), e2311148 (2024). https://doi.org/10.1002/adma.202311148
L. Zhang, M. Zhou, A. Wang, T. Zhang, Selective hydrogenation over supported metal catalysts: from nanops to single atoms. Chem. Rev. 120(2), 683–733 (2020). https://doi.org/10.1021/acs.chemrev.9b00230
X.-F. Yang, A. Wang, B. Qiao, J. Li, J. Liu et al., Single-atom catalysts: a new frontier in heterogeneous catalysis. Acc. Chem. Res. 46(8), 1740–1748 (2013). https://doi.org/10.1021/ar300361m
N. Kang, L. Liao, X. Zhang, Z. He, B. Yu et al., Engineering the axial coordination of cobalt single atom catalysts for efficient photocatalytic hydrogen evolution. Nano Res. 17(6), 5114–5121 (2024). https://doi.org/10.1007/s12274-024-6411-1
J.-B. Pan, B.-H. Wang, S. Shen, L. Chen, S.-F. Yin, Introducing bidirectional axial coordination into BiVO4 @metal phthalocyanine core-shell photoanodes for efficient water oxidation. Angew. Chem. Int. Ed. Engl. 62(38), e202307246 (2023). https://doi.org/10.1002/anie.202307246
D. Liu, X. Wan, J. Shui, Tailoring oxygen reduction reaction on M-N-C catalysts via axial coordination engineering. Small 20(50), e2406078 (2024). https://doi.org/10.1002/smll.202406078
W. Song, C. Xiao, J. Ding, Z. Huang, X. Yang et al., Review of carbon support coordination environments for single metal atom electrocatalysts (SACS). Adv. Mater. 36(1), e2301477 (2024). https://doi.org/10.1002/adma.202301477
L. Yin, M. Sun, S. Zhang, Y. Huang, B. Huang et al., Chlorine axial coordination activated lanthanum single atoms for efficient oxygen electroreduction with maximum utilization. Adv. Mater. 37(7), e2416387 (2025). https://doi.org/10.1002/adma.202416387
S. Ren, Y. Wang, L. Shi, X. Xu, S. Zhong et al., Transforming plastics to single atom catalysts for peroxymonosulfate activation: axial chloride coordination intensified electron transfer pathway. Adv. Mater. 37(8), e2415339 (2025). https://doi.org/10.1002/adma.202415339
Z. Zhang, Z. Feng, X. Zhao, D. Jean, Z. Yu et al., Functionalization and higher-order organization of liposomes with DNA nanostructures. Nat. Commun. 14(1), 5256 (2023). https://doi.org/10.1038/s41467-023-41013-2
C. Wang, X. Lan, L. Zhu, Y. Wang, X. Gao et al., Construction strategy of functionalized liposomes and multidimensional application. Small 20(25), e2309031 (2024). https://doi.org/10.1002/smll.202309031
V.P. Torchilin, Recent advances with liposomes as pharmaceutical carriers. Nat. Rev. Drug Discov. 4(2), 145–160 (2005). https://doi.org/10.1038/nrd1632
N. Filipczak, J. Pan, S.S.K. Yalamarty, V.P. Torchilin, Recent advancements in liposome technology. Adv. Drug Deliv. Rev. 156, 4–22 (2020). https://doi.org/10.1016/j.addr.2020.06.022
M. Lyu, M. Luo, J. Li, O.U. Akakuru, X. Fan et al., Personalized carbon monoxide-loaded biomimetic single-atom nanozyme for ferroptosis-enhanced FLASH radioimmunotherapy. Adv. Funct. Mater. 33(51), 2306930 (2023). https://doi.org/10.1002/adfm.202306930
X.T. Le, N.T. Nguyen, W.T. Lee, Y. Yang, H.-G. Choi et al., Peroxidase-mimicking iron-based single-atom upconversion photocatalyst for enhancing chemodynamic therapy. Adv. Funct. Mater. 34(34), 2401893 (2024). https://doi.org/10.1002/adfm.202401893
J.-H. Lee, Y. Shin, W. Lee, K. Whang, D. Kim et al., General and programmable synthesis of hybrid liposome/metal nanops. Sci. Adv. 2(12), e1601838 (2016). https://doi.org/10.1126/sciadv.1601838
H. Zhang, G. Liu, L. Shi, J. Ye, Single-atom catalysts: emerging multifunctional materials in heterogeneous catalysis. Adv. Energy Mater. 8, 1701343 (2018). https://doi.org/10.1002/aenm.201701343
Y. Zhang, J. Yang, R. Ge, J. Zhang, J.M. Cairney et al., The effect of coordination environment on the activity and selectivity of single-atom catalysts. Coord. Chem. Rev. 461, 214493 (2022). https://doi.org/10.1016/j.ccr.2022.214493
J. Chang, W. Jing, X. Yong, A. Cao, J. Yu et al., Synthesis of ultrahigh-metal-density single-atom catalysts via metal sulfide-mediated atomic trapping. Nat. Synth 3(11), 1427–1438 (2024). https://doi.org/10.1038/s44160-024-00607-4
D. Huang, L. Yin, X. Lu, S. Lin, Z. Niu et al., Directional electron transfer mechanisms with graphene quantum dots as the electron donor for photodecomposition of perfluorooctane sulfonate. Chem. Eng. J. 323, 406–414 (2017). https://doi.org/10.1016/j.cej.2017.04.124
H. Wei, K. Huang, D. Wang, R. Zhang, B. Ge et al., Iced photochemical reduction to synthesize atomically dispersed metals by suppressing nanocrystal growth. Nat. Commun. 8(1), 1490 (2017). https://doi.org/10.1038/s41467-017-01521-4
F. Maurer, J. Jelic, J. Wang, A. Gänzler, P. Dolcet et al., Tracking the formation, fate and consequence for catalytic activity of Pt single sites on CeO2. Nat. Catal. 3(10), 824–833 (2020). https://doi.org/10.1038/s41929-020-00508-7
X. Liu, X. Song, G. Jiang, L. Tao, Z. Jin et al., Pt single-atom collaborate with Pt atom-clusters by an in-situ confined strategy for accelerating electrocatalytic hydrogen evolution. Chem. Eng. J. 481, 148430 (2024). https://doi.org/10.1016/j.cej.2023.148430
Z. Xu, J. Jiang, Y. Li, T. Hu, J. Gu et al., Shape-regulated photothermal-catalytic tumor therapy using Polydopamine@Pt nanozymes with the elicitation of an immune response. Small 20(20), e2309096 (2024). https://doi.org/10.1002/smll.202309096
K. Deng, H. Hu, Y. Li, X. Li, H. Deng et al., Mechanistic investigation and dual-mode colorimetric-chemiluminescent detection of glyphosate based on the specific inhibition of Fe3O4@Cu nanozyme peroxidase-like activity. Food Chem. 443, 138501 (2024). https://doi.org/10.1016/j.foodchem.2024.138501
B.H.J. Hofstee, On the evaluation of the constants Vm and KM in enzyme reactions. Science 116(3013), 329–331 (1952). https://doi.org/10.1126/science.116.3013.329
M. Sela, M. Poley, P. Mora-Raimundo, S. Kagan, A. Avital et al., Brain-targeted liposomes loaded with monoclonal antibodies reduce alpha-synuclein aggregation and improve behavioral symptoms in Parkinson’s disease. Adv. Mater. 35(51), e2304654 (2023). https://doi.org/10.1002/adma.202304654
A.B. Ganganboina, A.D. Chowdhury, I.M. Khoris, F. Nasrin, K. Takemura et al., Dual modality sensor using liposome-based signal amplification technique for ultrasensitive norovirus detection. Biosens. Bioelectron. 157, 112169 (2020). https://doi.org/10.1016/j.bios.2020.112169
G. Seo, G. Lee, M.J. Kim, S.-H. Baek, M. Choi et al., Rapid detection of COVID-19 causative virus (SARS-CoV-2) in human nasopharyngeal swab specimens using field-effect transistor-based biosensor. ACS Nano 14(4), 5135–5142 (2020). https://doi.org/10.1021/acsnano.0c02823
J. Shang, Y. Wang, Y. Pang, M. Yu, Y. Wang et al., Point-of-care diagnosis of respiratory viruses at single-nucleotide resolution with an autocatalytic rolling circle amplification system. Angew. Chem. Int. Ed. 64(52), e18925 (2025). https://doi.org/10.1002/anie.202518925
A. Piriya V S, P. Joseph, K. Daniel S C G, S. Lakshmanan, T. Kinoshita et al., Colorimetric sensors for rapid detection of various analytes. Mater. Sci. Eng. C Mater. Biol. Appl. 78, 1231–1245 (2017). https://doi.org/10.1016/j.msec.2017.05.018
R. Cui, H. Tang, Q. Huang, T. Ye, J. Chen et al., AI-assisted smartphone-based colorimetric biosensor for visualized, rapid and sensitive detection of pathogenic bacteria. Biosens. Bioelectron. 259, 116369 (2024). https://doi.org/10.1016/j.bios.2024.116369
S. Wang, Y. Zhu, Z. Zhou, Y. Luo, Y. Huang et al., Integrated ultrasound-enrichment and machine learning in colorimetric lateral flow assay for accurate and sensitive clinical Alzheimer’s biomarker diagnosis. Adv. Sci. 11(42), 2406196 (2024). https://doi.org/10.1002/advs.202406196
T. He, Z. Zhang, H. Zhang, Z. Zhang, J. Xie et al., Bag of tricks for image classification with convolutional neural networks. 2019 IEEE/CVF Conference on Computer Vision and Pattern Recognition (CVPR)., 558–567. IEEE (2020). https://doi.org/10.1109/CVPR.2019.00065